Abstract
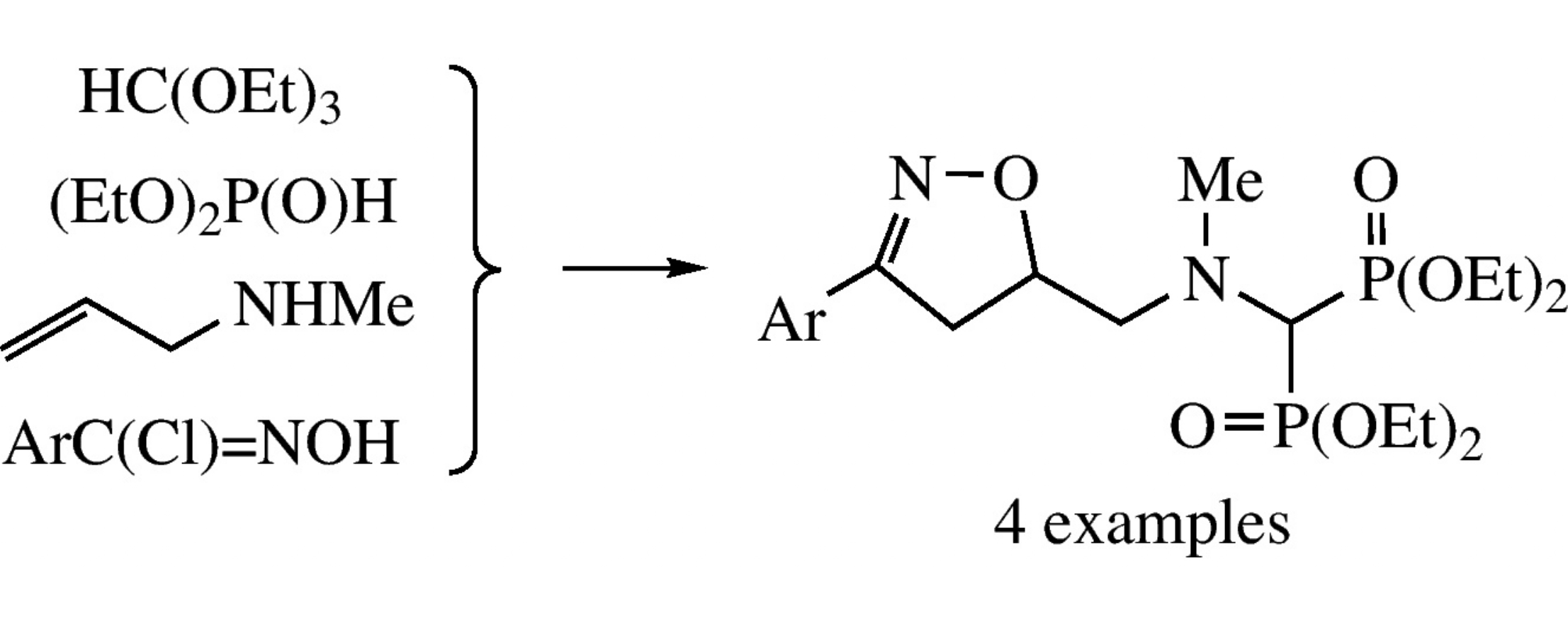
Three-component reaction between N-allyl-N-methylamine, triethyl orthoformate and diethyl phosphite affords N-[bis(diethoxyphosphoryl)methyl]-N-methyl-3-arylprop-3-en-1-amine whose cycloaddition with nitrile N-oxides gives 5-{N-[bis(diethoxyphosphoryl)methyl]-N-methylamino}methyl-4,5-dihydroisoxazoles.
References
1.
Romanenko V.D., Kukhar V.P.
Arkivoc,
2012
2.

Zhang S., Gangal G., Uludağ H.
Chemical Society Reviews,
2007
3.

RUSSELL R.G.
Annals of the New York Academy of Sciences,
2006
4.

Fleisch H.
Breast Cancer Research,
2001
5.

Morley A.D., Cook A., King S., Roberts B., Lever S., Weaver R., MacDonald C., Unitt J., Fagura M., Phillips T., Lewis R., Wenlock M.
Bioorganic and Medicinal Chemistry Letters,
2011
6.

7.
T. M. Balashova and I. D. Kolpakova, in Metody polucheniya khimicheskikh reaktivov i preparatov (Methods of Obtaining of Chemical Reagents and Preparations), 1973, vol. 25, p. 11 (in Russian).
8.

Pl�ger W., Schindler N., Wollmann K., Worms K.H.
Zeitschrift fur Anorganische und Allgemeine Chemie,
1972
9.

Prishchenko A.A., Livantsov M.V., Novikova O.P., Livantsova L.I.
Russian Journal of General Chemistry,
2009
10.
10.1016/j.mencom.2015.05.027_sbref0040a
Maier
Elem.,
1981
11.

MIMURA M., HAYASHIDA M., NOMIYAMA K., IKEGAMI S., IIDA Y., TAMURA M., HIYAMA Y., OHISHI Y.
Chemical and Pharmaceutical Bulletin,
2011
12.

Jäger V., Colinas P.A.
Cumulative Index of Heterocyclic Systems,
2002
13.

Liu K., Shelton B.R., Howe R.K.
Journal of Organic Chemistry,
1980
14.

Olsson T., Stern K., Sundell S.
Journal of Organic Chemistry,
1988
15.

Lazrek H.B., Rochdi A., Khaider H., Barascut J.-., Imbach J.-., Balzarini J., Witvrouw M., Pannecouque C., De Clercq E.
Tetrahedron,
1998