Abstract
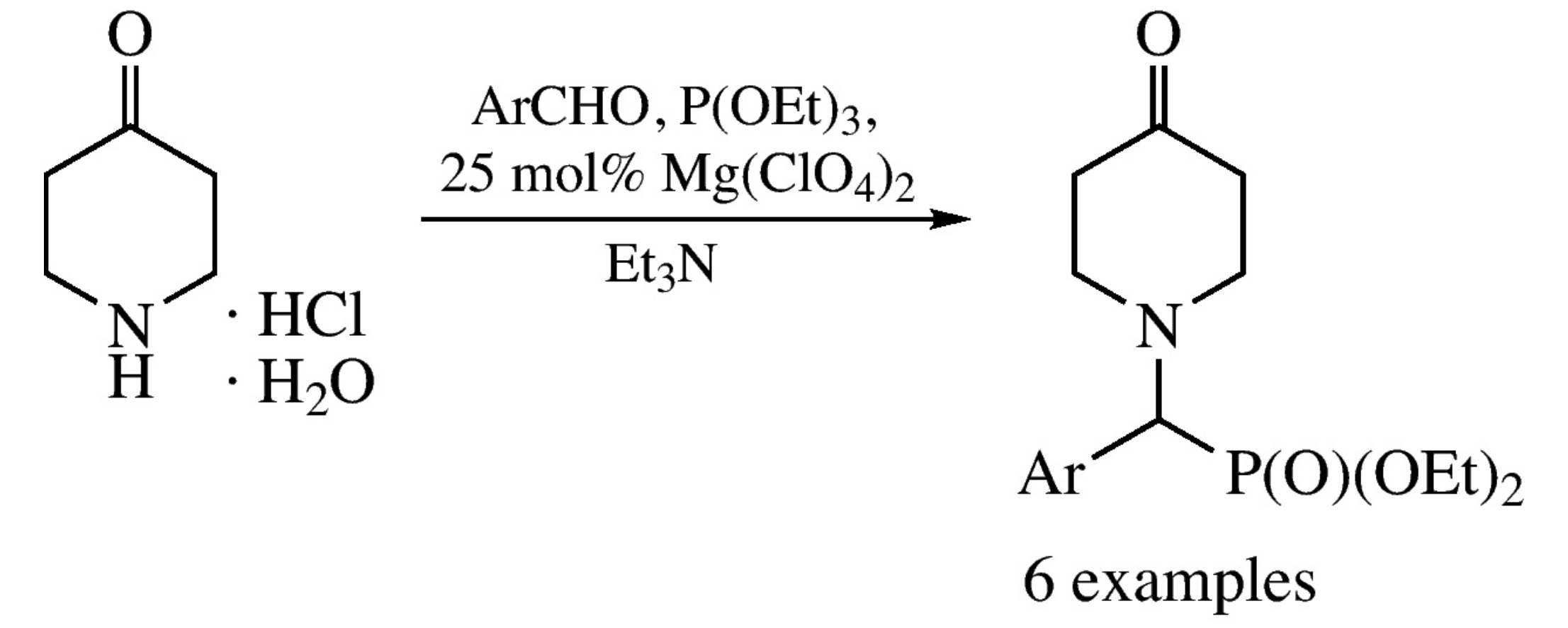
The Kabachnik–Fields reaction of 4-piperidone hydrochloride monohydrate, an aromatic aldehyde and triethyl phosphite in the presence of triethylamine and magnesium perchlorate affords diethyl (aryl)(4-oxopiperidin-1-yl)methylphosphonates in moderate yields.
References
1.

Kant Sahu S., Kumar Dubey B., C. Tripathi A., Koshy M., K. Saraf S.
Mini-Reviews in Medicinal Chemistry,
2013
2.

Kurbat N.M., Praliev K.D., Salita T.A., Yu V.K., Verina E.L.
Pharmaceutical Chemistry Journal,
1991
3.

Das U., Sharma R., Dimmock J.
Current Medicinal Chemistry,
2009
4.

Krise J.P., Stella V.J.
Advanced Drug Delivery Reviews,
1996
5.

Hecker S.J., Erion M.D.
Journal of Medicinal Chemistry,
2008
6.

M. Huttunen K., Rautio J.
Current Topics in Medicinal Chemistry,
2011
7.

Baylis E.K., Campbell C.D., Dingwall J.G.
Journal of the Chemical Society Perkin Transactions 1,
1984
8.
10.1016/j.mencom.2015.05.026_sbref0025b
Kafarski
Elem.,
1991
9.

Mucha A., Kafarski P., Berlicki Ł.
Journal of Medicinal Chemistry,
2011
10.

Makarov M.V., Rybalkina E.Y., Röschenthaler G., Short K.W., Timofeeva T.V., Odinets I.L.
European Journal of Medicinal Chemistry,
2009
11.
(a) M. I. Kabachnik and T. Ya. Medved, Dokl. Akad. Nauk SSSR, 1952, 83, 689 (Chem. Abstr., 1953, 47, 2724b); (b) E. K. Fields, J. Am. Chem. Soc., 1952, 74, 1528.
12.

Cherkasov R.A., Galkin V.I.
Russian Chemical Reviews,
1998
13.

Keglevich G., Szekrenyi A.
Letters in Organic Chemistry,
2008
14.

Keglevich G., Bálint E., Kangyal R., Bálint M., Milen M.
Heteroatom Chemistry,
2014
15.

Vahdat S.M., Baharfar R., Tajbakhsh M., Heydari A., Baghbanian S.M., Khaksar S.
Tetrahedron Letters,
2008
16.
10.1016/j.mencom.2015.05.026_sbref0055b
Tajbakhsh
Synthesis,
2008
17.

Maghsoodlou M.T., Habibi-Khorassani S.M., Heydari R., Hazeri N., Sajadikhah S.S., Rostamizadeh M.
Arabian Journal of Chemistry,
2011
18.

Bhagat S., Chakraborti A.K.
Journal of Organic Chemistry,
2007