Abstract
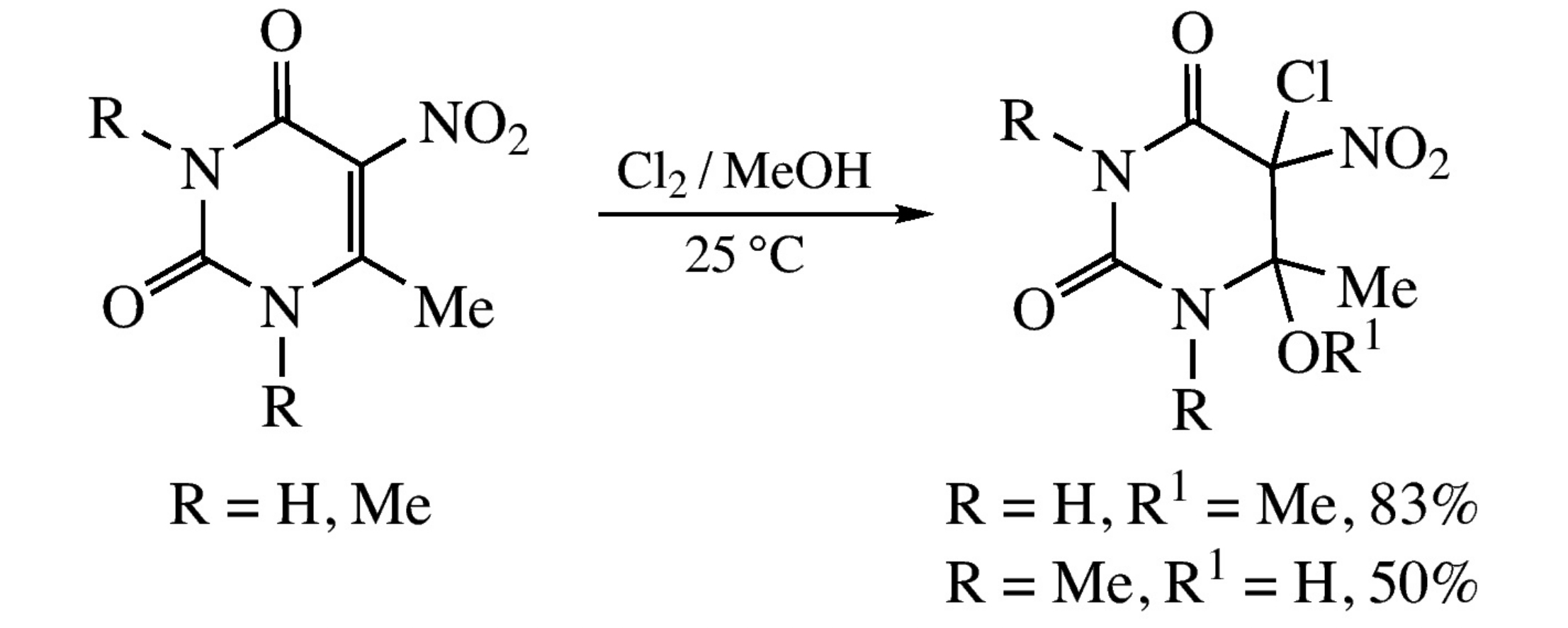
The chlorination of 6-methyl-5-nitrouracil with chlorine in methanol results in a two-component mixture of 5-chloro-6-methyl-6-methoxy-5-nitro-5,6-dihydrouracils, while the chlorination of 5-nitro-1,3,6-trimethyluracil under similar conditions gives a mixture of 5-chloro-6-hydroxy-5-nitro-1,3,6-trimethyl-5,6-dihydrouracils. The structures of the chlorination products were suggested based on quantum-chemical calculations
References
1.
10.1016/j.mencom.2015.05.022_bib0005
Ovchinnikov Yu.
Bioorganicheskaya khimiya (Bioorganic Chemistry),
1987
2.
![Synthesis and Antiviral Activity of 1-{[2-(Phenoxy)ethoxy]methyl}uracil Derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Novikov M.S., Ozerov A.A., Orlova Y.A., Buckheit R.W.
Chemistry of Heterocyclic Compounds,
2005
3.

Novikov M.S., Ozerov A.A., Brel' A.K., Solodunova G.N., Ozerova T.P.
Chemistry of Heterocyclic Compounds,
1996
4.

Ozerov A.A., Novikov M.S., Brel' A.K., Andreeva O.T., Vladykov G.V., Boreko E.I., Korobchenko L.V., Vervetchenko S.G.
Pharmaceutical Chemistry Journal,
1991
5.

Structure and activity relationships of novel uracil derivatives as topical anti-inflammatory agents
Isobe Y., Tobe M., Inoue Y., Isobe M., Tsuchiya M., Hayashi H.
Bioorganic and Medicinal Chemistry,
2003
6.
10.1016/j.mencom.2015.05.022_bib0030
Lazareva
Med. Vestn. Bashkortostana,
2007
7.
V. A. Myshkin and A. B. Bakirov, Oksimetiluratsil. Ocherki eksperimental’noi farmakologii (Oxymethyluracil. Essays of Experimental Pharmacology), DAR, Ufa, 2001, p. 218 (in Russian).
8.
10.1016/j.mencom.2015.05.022_bib0040
Gimadieva
Bash. Khim. Zh.,
2007
9.

Ren H., Yang Y., Lin J., Qi Y., Zhang Y.
Frontiers of Chemistry in China,
2008
10.

Kasradze V.G., Ignatyeva I.B., Khusnutdinov R.A., Suponitskii K.Y., Antipin M.Y., Yunusov M.S.
Chemistry of Heterocyclic Compounds,
2012
11.

Chernikova I.B., Khursan S.L., Spirikhin L.V., Yunusov M.S.
Russian Chemical Bulletin,
2013
12.
10.1016/j.mencom.2015.05.022_bib0060
Fattakhov Kh.
PhD Thesis, Ufa,
2011
13.
Nelson R.D., Lide D.R., Maryott A.A.
2016
14.
J. Mathieu and R. Panico, Kurs teoreticheskikh osnov organicheskoi khimii (Course of Theoretical Bases of Organic Chemistry), Mir, Moscow, 1975, p. 80 (in Russian).
15.
10.1016/j.mencom.2015.05.022_bib0075
Frisch
Gaussian 09, Revision C.1, Gaussian, Inc., Wallingford CT,
2009
16.
G. A. Zhurko, ChemCraft, Version 1.6, http://www.chemcraftprog.com.
17.

Becke A.D.
Journal of Chemical Physics,
1993
18.

Lee C., Yang W., Parr R.G.
Physical Review B,
1988
19.

McLean A.D., Chandler G.S.
Journal of Chemical Physics,
1980
20.

Krishnan R., Binkley J.S., Seeger R., Pople J.A.
Journal of Chemical Physics,
1980
21.

Tomasi J., Mennucci B., Cammi R.
Chemical Reviews,
2005
22.

Reed A.E., Curtiss L.A., Weinhold F.
Chemical Reviews,
1988
23.
E. Kirby, Anomernyi effekt kislorodsoderzhashchikh soedinenii (Anomeric Effect of Oxygen-containing Compounds), Mir, Moscow, 1985, p. 171 (in Russian).