Abstract
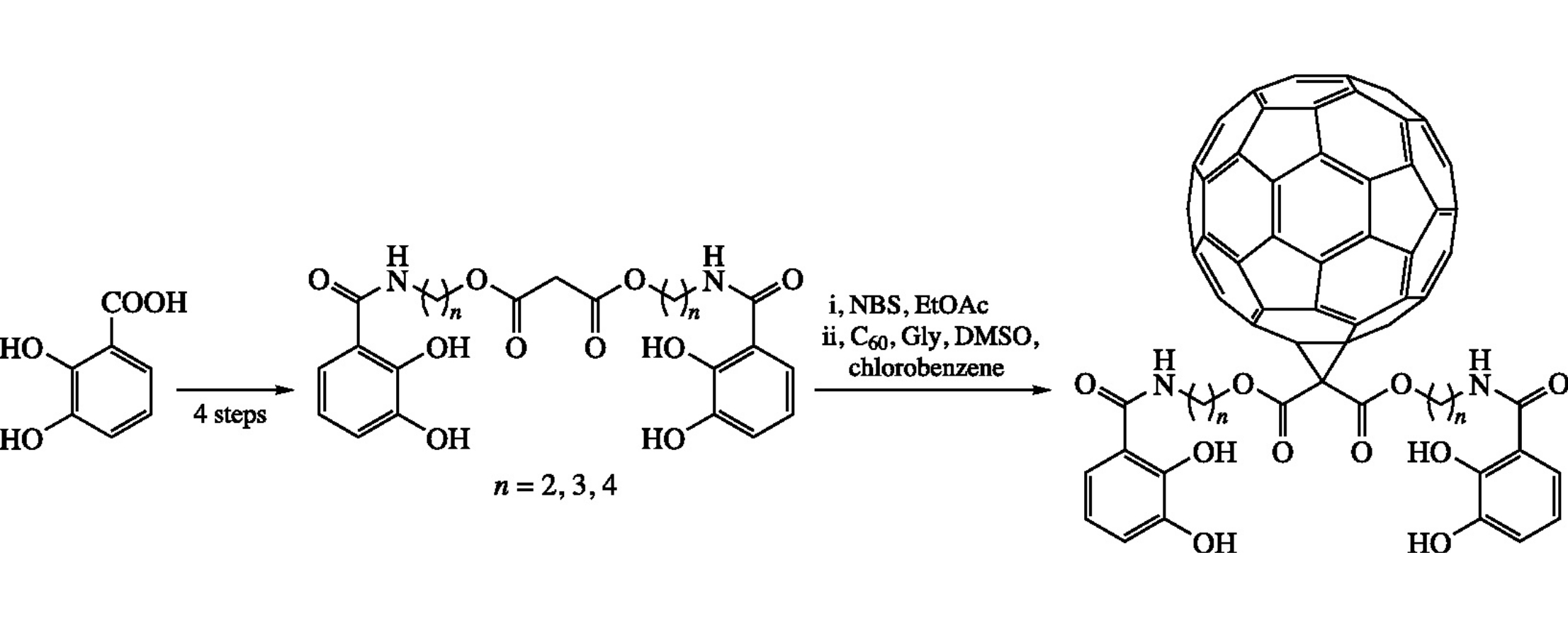
The new C60-Ih fullerene-based catechol amide derivatives C60{C[COO(CH2)nCAM]2} [n = 2, 3 or 4; CAM = 2,3-Ph(OH)2CONH] were prepared using a modified Bingel-type reaction of C60-Ih and characterized by variable-temperature 1H NMR spectroscopy.
References
1.
P. Galle, Toxiques Nucléaires, Masson, Paris, 1997, pp. 185-209.
2.

Brugge D., deLemos J.L., Oldmixon B.
Reviews on Environmental Health,
2005
3.
10.1016/j.mencom.2015.05.015_bib0015
Durbin
2006
4.
10.1016/j.mencom.2015.05.015_bib0020
Pellmar
Neurotoxicology,
1999
5.

Periyakaruppan A., Kumar F., Sarkar S., Sharma C.S., Ramesh G.T.
Archives of Toxicology,
2006
6.

Sylwester E.R., Allen P.G., Dharmawardana U.R., Sutton M.
Inorganic Chemistry,
2001
7.

Gorden A.E., Xu J., Raymond K.N., Durbin P.
Chemical Reviews,
2003
8.

Leydier A., Lecerclé D., Pellet-Rostaing S., Favre-Reguillon A., Taran F., Lemaire M.
Tetrahedron,
2008
9.

Leydier A., Lecerclé D., Pellet-Rostaing S., Favre-Réguillon A., Taran F., Lemaire M.
Tetrahedron,
2008
10.
C. D. Gutsche, in Calixarenes Revisited. Monographs in Supramolecular Chemistry, ed. J. F. Stoddart, The Royal Society of Chemistry, Cambridge, 1998, vol. 6.
11.

Leydier A., Lecerclé D., Pellet-Rostaing S., Favre-Reguillon A., Taran F., Lemaire M.
Tetrahedron Letters,
2011
12.

Leydier A., Lin Y., Arrachart G., Turgis R., Lecerclé D., Favre-Reguillon A., Taran F., Lemaire M., Pellet-Rostaing S.
Tetrahedron,
2012
13.
10.1016/j.mencom.2015.05.015_bib0065
Andrievsky
Med.,
2009
14.

Krusic P.J., Wasserman E., Keizer P.N., Morton J.R., Preston K.F.
Science,
1991
15.

Dugan L.L., Lovett E.G., Quick K.L., Lotharius J., Lin T.T., O'Malley K.L.
Parkinsonism and Related Disorders,
2001
16.

Xiao L., Takada H., Maeda K., Haramoto M., Miwa N.
Biomedicine and Pharmacotherapy,
2005
17.

Yin J., Lao F., Fu P.P., Wamer W.G., Zhao Y., Wang P.C., Qiu Y., Sun B., Xing G., Dong J., Liang X., Chen C.
Biomaterials,
2009
18.

Xiao L., Takada H., Gan X.H., Miwa N.
Bioorganic and Medicinal Chemistry Letters,
2006
19.
Kornev A.B., Peregudov A.S., Martynenko V.M., Guseva G.V., Sashenkova T.E., Rybkin A.Y., Faingold I.I., Mishchenko D.V., Kotelnikova R.A., Konovalova N.P., Balzarini J., Troshin P.A.
Mendeleev Communications,
2013
20.

Laursen B., Denieul M., Skrydstrup T.
Tetrahedron,
2002
21.

Gardner R.A., Kinkade R., Wang C., Phanstiel
Journal of Organic Chemistry,
2004
22.

Camps X., Hirsch A.
Journal of the Chemical Society Perkin Transactions 1,
1997
23.

Hummelen J.C., Knight B.W., LePeq F., Wudl F., Yao J., Wilkins C.L.
Journal of Organic Chemistry,
1995
24.

Scharff P., Risch K., Carta-Abelmann L., Dmytruk I.M., Bilyi M.M., Golub O.A., Khavryuchenko A.V., Buzaneva E.V., Aksenov V.L., Avdeev M.V., Prylutskyy Y.I., Durov S.S.
Carbon,
2004
25.

Maggini M., Scorrano G., Prato M.
Journal of the American Chemical Society,
1993
26.

Diederich F., Isaacs L., Philp D.
Chemical Society Reviews,
1994
27.

Borsato G., Negra F.D., Gasparrini F., Misiti D., Lucchini V., Possamai G., Villani C., Zambon A.
Journal of Organic Chemistry,
2004
28.
![[60]Fullerene as a Substituent](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bagno A., Claeson S., Maggini M., Martini M.L., Prato M., Scorrano G.
Chemistry - A European Journal,
2002
29.

Wharton T., Wilson L.J.
Bioorganic and Medicinal Chemistry,
2002
30.

Bradamante S., Vittadini G.
Magnetic Resonance in Chemistry,
1987
31.
![Efficient cyclopropanation of [60]fullerene starting from bromo-substituted active methylene compounds without using a basic catalyst](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Jin B., Shen J., Peng R., Zheng R., Chu S.
Tetrahedron Letters,
2014
32.

Photochemical α-bromination of ketones using N-bromosuccinimide: a simple, mild and efficient method
Arbuj S.S., Waghmode S.B., Ramaswamy A.V.
Tetrahedron Letters,
2007