Abstract
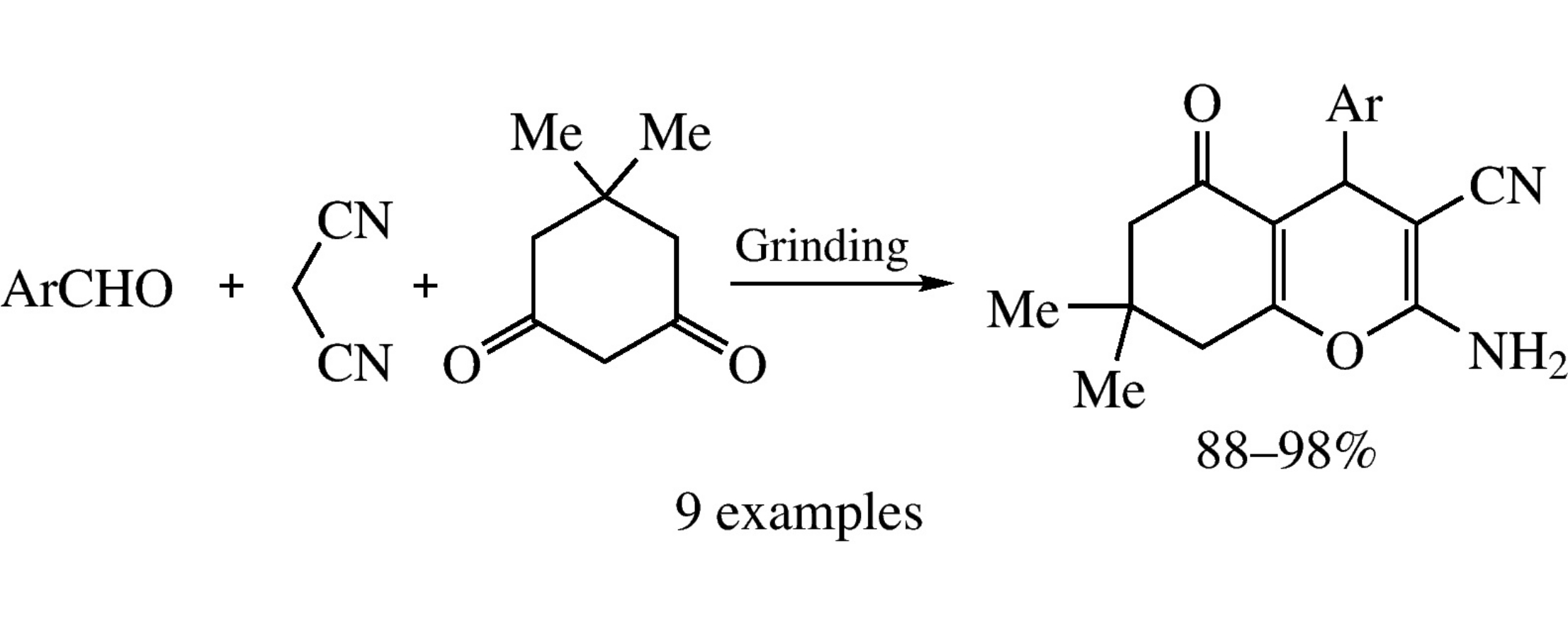
Non-catalytic solvent-free assembling of aldehydes, dimedone and malononitrile at ambient temperature affords substituted 5,6,7,8-tetrahydro-4H-chromenes in 88–98% yields.
References
1.
Multicomponent Reactions, eds. J. Zhu and H. Bienayme, Wiley-VCH, Weinheim, 2005.
2.

D'Souza D.M., Müller T.J.
Chemical Society Reviews,
2007
3.
Synthesis of Heterocycles via Multicomponent Reactions I, eds. R. V. A. Orru and E. Ruijter, Springer, New York, 2010.
4.

Weber L.
Drug Discovery Today,
2002
5.

Dömling A.
Current Opinion in Chemical Biology,
2002
6.
Drug Design, ed. G. Klebe, Springer, Berlin, 2013.
7.

Skommer J., Wlodkowic D., Mättö M., Eray M., Pelkonen J.
Leukemia Research,
2006
8.

Yu N., Aramini J.M., Germann M.W., Huang Z.
Tetrahedron Letters,
2000
9.

Bonsignore L., Loy G., Secci D., Calignano A.
European Journal of Medicinal Chemistry,
1993
10.

Parmar V.S., Jain S.C., Bisht K.S., Jain R., Taneja P., Jha A., Tyagi O.D., Prasad A.K., Wengel J., Olsen C.E., Boll P.M.
Phytochemistry,
1997
11.

Polyakov V.V.
Chemistry of Natural Compounds,
1999
12.
10.1016/j.mencom.2015.05.008_bib0060
Konkoy
PCT Int. Appl. WO 0075123,
2000
13.
10.1016/j.mencom.2015.05.008_bib0065
Abdel-Latif
J. Chem. Res. Miniprint,
1995
14.
10.1016/j.mencom.2015.05.008_bib0070
Hassanien
J. Indian Chem. Soc.,
1999
15.
![One-step synthesis of substituted 2-amino-5,6,7,8-tetrahydro-4H-benzo[b]pyrans. Molecular and crystal structure of 2-amino-3-(2-methoxyethoxycarbonyl)-4-(2-nitrophenyl)-5-oxo-5,6,7,8-tetrahydro-4H-benzo[b]pyran](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Shestopalov A.M., Emelianova Y.M., Nesterov V.N.
Russian Chemical Bulletin,
2003
16.

Jin T., Wang A., Wang X., Zhang J., Li T.
Synlett,
2004
17.

Gao S., Tsai C.H., Tseng C., Yao C.
Tetrahedron,
2008
18.
![(S)-Proline as a Neutral and Efficient Catalyst for the One-Pot Synthesis of Tetrahydrobenzo[b]pyran Derivatives in Aqueous Media](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Balalaie S., Bararjanian M., Amani A.M., Movassagh B.
Synlett,
2006
19.

Dekamin M.G., Eslami M., Maleki A.
Tetrahedron,
2013
20.

Baghbanian S.M., Rezaei N., Tashakkorian H.
Green Chemistry,
2013
21.

Safaei H.R., Shekouhy M., Rahmanpur S., Shirinfeshan A.
Green Chemistry,
2012
22.
![Electrogenerated base-promoted synthesis of tetrahydrobenzo[b]pyran derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fotouhi L., Heravi M.M., Fatehi A., Bakhtiari K.
Tetrahedron Letters,
2007
23.

Elinson M.N., Dorofeev A.S., Feducovich S.K., Gorbunov S.V., Nasybullin R.F., Miloserdov F.M., Nikishin G.I.
European Journal of Organic Chemistry,
2006
24.
![Sodium bromide catalysed one-pot synthesis of tetrahydrobenzo[b]pyrans via a three-component cyclocondensation under microwave irradiation and solvent free conditions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
25.

Kaupp G., Reza Naimi-Jamal M., Schmeyers J.
Tetrahedron,
2003
26.

Wender P.A.
Natural Product Reports,
2014
27.

Elinson M.N., Ilovaisky A.I., Merkulova V.M., Belyakov P.A., Chizhov A.O., Nikishin G.I.
Tetrahedron,
2010
28.
Elinson M.N., Medvedev M.G., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2013
29.

Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Zaimovskaya T.A., Egorov M.P.
Monatshefte fur Chemie,
2014
30.

Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Egorov M.P.
Comptes Rendus Chimie,
2013
31.
![Solvent-free multicomponent assembling of aldehydes, N,N′-dialkyl barbiturates and malononitrile: fast and efficient approach to pyrano[2,3-d]pyrimidines](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Elinson M.N., Ryzhkov F.V., Merkulova V.M., Ilovaisky A.I., Nikishin G.I.
Heterocyclic Communications,
2014
32.
![Solvent-free and ‘on-water’ multicomponent assembling of aldehydes, 3-methyl-2-pyrazoline-5-one, and malononitrile: fast and efficient approach to medicinally relevant pyrano[2,3-c]pyrazole scaffold](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Zaimovskaya T.A., Nikishin G.I.
Monatshefte fur Chemie,
2015
33.

Elinson M.N., Ilovaisky A.I., Merkulova V.M., Belyakov P.A., Barba F., Batanero B.
Tetrahedron,
2012
34.
Elinson M.N., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2012
35.
Elinson M.N., Sokolova O.O., Nasybullin R.F., Zaimovskaya T.A., Egorov M.P.
Mendeleev Communications,
2015
36.

37.
![Stereoselective electrocatalytic transformations of malononitrile and aromatic aldehydes into (1R,5S,6R)*-4,4-dialkoxy-2-amino-6-aryl-1,5-dicyano-3-azabicyclo[3.1.0]hex-2-enes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Feducovich K., Zaimovskaya T.A., Vereshchagin A.N., Nikishin G.I.
Russian Chemical Bulletin,
2005
38.

Elinson M.N., Vereshchagin A.N., Feducovich S.K., Zaimovskaya T.A., Starikova Z.A., Belyakov P.A., Nikishin G.I.
Tetrahedron Letters,
2007
39.

Patai S., Israeli Y.
Journal of the Chemical Society (Resumed),
1960