Abstract
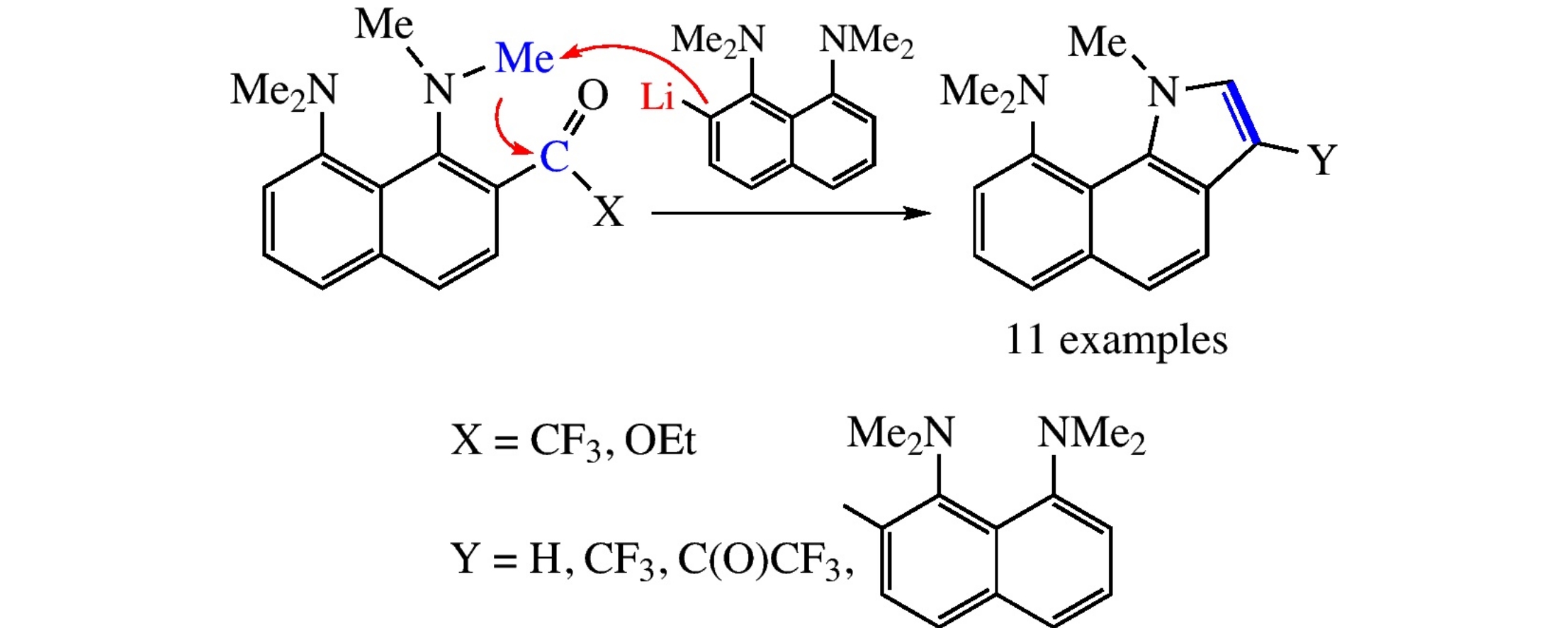
1,8-Bis(dimethylamino)naphthalenes bearing 2-positioned trifluoroacetyl or ethoxycarbonyl group on treatment with 2-lithio-1,8-bis-dimethylamino)naphthalene undergo base-promoted transformation into benzo[g]indole derivatives in small to moderate yield, representing previously unknown mode of the pyrrole ring closure which proceeds via deprotonation of the NMe group.
References
1.
10.1016/j.mencom.2015.05.007_sbref0005a
Alder
J. Chem. Soc., Chem. Commun.,
1968
2.

Hibbert F.
Journal of the Chemical Society Perkin Transactions 2,
1974
3.
10.1016/j.mencom.2015.05.007_bib0010
Pozharskii
The Chemistry of Anilines,
2007
4.
10.1016/j.mencom.2015.05.007_bib0015
Pozharskii
Russ. J. Org. Chem.,
1996
5.

Katritzky A.R., Qi M.
Tetrahedron,
1998
6.

Pozharskii A.F., Degtyarev A.V., Ryabtsova O.V., Ozeryanskii V.A., Kletskii M.E., Starikova Z.A., Sobczyk L., Filarowski A.
Journal of Organic Chemistry,
2007
7.

Farrer N.J., McDonald R., McIndoe J.S.
Dalton Transactions,
2006
8.

Pozharskii A.F., Tregub N.G., Vistorobskii N.V., Romanova E.Y., Starikova Z.A., Yanovsky A.I.
Russian Chemical Bulletin,
1999
9.
10.1016/j.mencom.2015.05.007_bib0040
Okada
Book of Abstracts. Congress of Heterocyclic Chemistry (Japan),
2003
10.

Pozharskii A.F., Degtyarev A.V., Ozeryanskii V.A., Ryabtsova O.V., Starikova Z.A., Borodkin G.S.
Journal of Organic Chemistry,
2010
11.
10.1016/j.mencom.2015.05.007_sbref0050a
Sundberg
1984
12.
10.1016/j.mencom.2015.05.007_sbref0050b
Sundberg
1996
13.
10.1016/j.mencom.2015.05.007_sbref0050c
Bergman
2008
14.

Boiko L.Z., Sorokin V.I., Filatova E.A., Starikova Z.A., Ozeryanskii V.A., Pozharskii A.F.
Journal of Molecular Structure,
2011