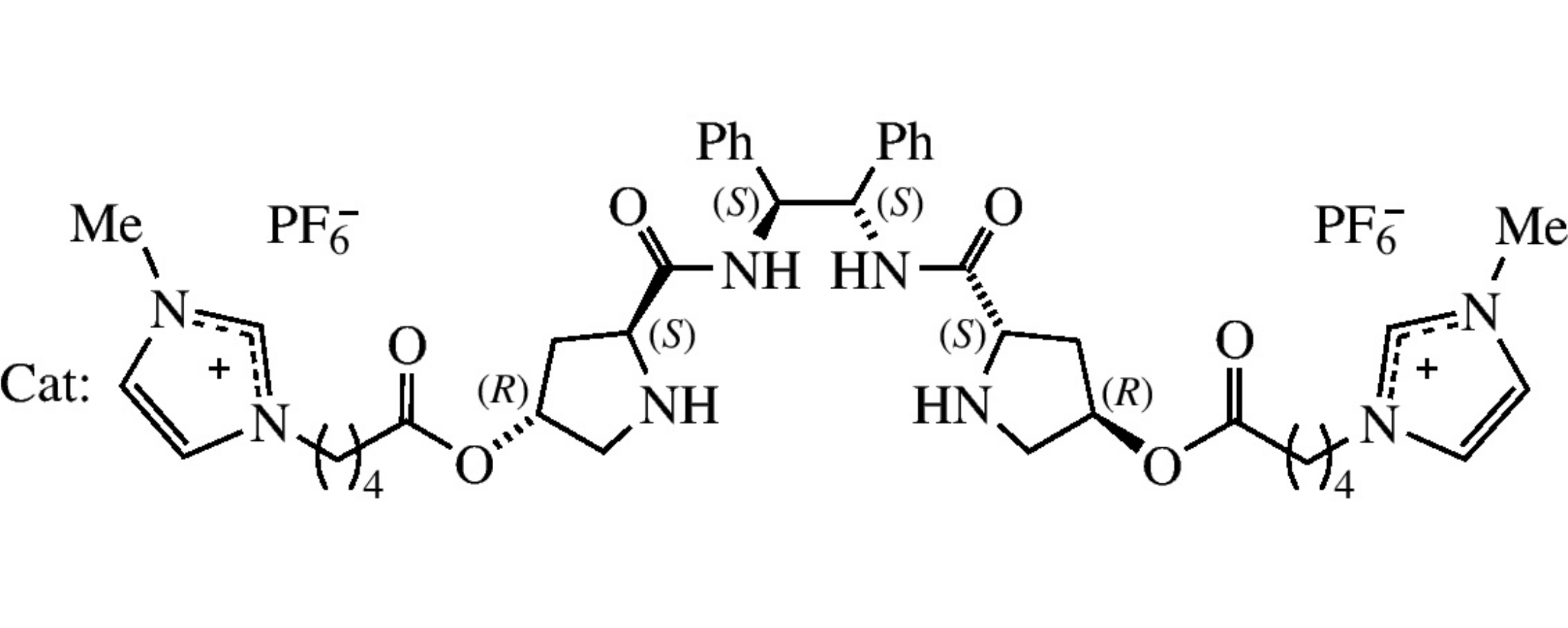
Abstract
Ionic liquid-supported (1S,2S)- and (1R,2R)-1,2-bis[(S)-prolinamido]-1,2-diphenylethanes act as organocatalysts in asymmetric aldol reactions of acetone with α-keto esters or trifluoroacetophenone providing high yields and from moderate to high enantioselectivity. Recycling of the catalyst with a gradual decrease in conversion and ee values is possible.
References
1.
Science of Synthesis: Asymmetric Organocatalysis, eds. B. List and K. Maruoka, Thieme, Stuttgart, 2012.
2.
Comprehensive Enantioselective Organocatalysis: Catalysts, Reactions, and Applications, ed. P. I. Dalco, Wiley-VCH, Weinheim, 2013.
3.

Marqués-López E., Herrera R.P., Christmann M.
Natural Product Reports,
2010
4.

Melchiorre P., Marigo M., Carlone A., Bartoli G.
Angewandte Chemie - International Edition,
2008
5.
10.1016/j.mencom.2015.05.002_sbref0010c
Rueping
Green Chem.,
2011
6.

Kristensen T.E., Hansen T.
European Journal of Organic Chemistry,
2010
7.
10.1016/j.mencom.2015.05.002_sbref0015b
Scheffler
Synlett,
2011
8.

Siyutkin D.E., Kucherenko A.S., Zlotin S.G.
2013
9.

Ni B., Headley A.
Chemistry - A European Journal,
2010
10.

Kucherenko A.S., Siyutkin D.E., Maltsev O.V., Kochetkov S.V., Zlotin S.G.
Russian Chemical Bulletin,
2012
11.

Šebesta R., Kmentová I., Toma Š.
Green Chemistry,
2008
12.
10.1016/j.mencom.2015.05.002_sbref0020e
Lombardo
Chem Cat Chem,
2010
13.

Brozinski H.L., Delaney J.P., Henderson L.C.
Australian Journal of Chemistry,
2013
14.

Miao W., Chan T.H.
Advanced Synthesis and Catalysis,
2006
15.

Gruttadauria M., Giacalone F., Noto R.
Chemical Society Reviews,
2008
16.

Siyutkin D.E., Kucherenko A.S., Struchkova M.I., Zlotin S.G.
Tetrahedron Letters,
2008
17.

Trindade A.F., Gois P.M., Afonso C.A.
Chemical Reviews,
2009
18.

Siyutkin D.E., Kucherenko A.S., Zlotin S.G.
Tetrahedron,
2010
19.

Larionova N.A., Kucherenko A.S., Siyutkin D.E., Zlotin S.G.
Tetrahedron,
2011
20.

Qiao Y., Headley A.
Catalysts,
2013
21.

SRIVASTAVA V.
Journal of Chemical Sciences,
2013
22.

Bañón-Caballero A., Guillena G., Nájera C., Faggi E., Sebastián R.M., Vallribera A.
Tetrahedron,
2013
23.

Pedrosa R., Andrés J.M., Manzano R., Pérez-López C.
Tetrahedron Letters,
2013
24.

Senanayake C.H., Fang K., Grover P., Bakale R.P., Vandenbossche C.P., Wald S.A.
Tetrahedron Letters,
1999
25.

Grover P.T., Bhongle N.N., Wald S.A., Senanayake C.H.
Journal of Organic Chemistry,
2000
26.

Masumoto S., Suzuki M., Kanai M., Shibasaki M.
Tetrahedron Letters,
2002
27.

Gupta P., Fernandes R.A., Kumar P.
Tetrahedron Letters,
2003
28.

Masumoto S., Suzuki M., Kanai M., Shibasaki M.
Tetrahedron,
2004
29.

Tokuda O., Kano T., Gao W., Ikemoto T., Maruoka K.
Organic Letters,
2005
30.

Wang F., Xiong Y., Liu X., Feng X.
Advanced Synthesis and Catalysis,
2007
31.

Liu J., Yang Z., Wang Z., Wang F., Chen X., Liu X., Feng X., Su Z., Hu C.
Journal of the American Chemical Society,
2008
32.

Liu Q., Wang X., Luo S., Zheng B., Qin D.
Tetrahedron Letters,
2008
33.
10.1016/j.mencom.2015.05.002_sbref0040d
Raj
Catal,
2009
34.

Jiang Z., Lu Y.
Tetrahedron Letters,
2010
35.

Zhu X., Lin A., Fang L., Li W., Zhu C., Cheng Y.
Chemistry - A European Journal,
2011
36.

Viózquez S.F., Bañón-Caballero A., Guillena G., Nájera C., Gómez-Bengoa E.
Organic and Biomolecular Chemistry,
2012
37.

Mikami K., Kawakami Y., Akiyama K., Aikawa K.
Journal of the American Chemical Society,
2007
38.

Tang Z., Cun L., Cui X., Mi A., Jiang Y., Gong L.
Organic Letters,
2006
39.

Kokotos C.G.
Journal of Organic Chemistry,
2012
40.

Xu X., Tang Z., Wang Y., Luo S., Cun L., Gong L.
Journal of Organic Chemistry,
2007
41.
42.
43.

Müller K., Faeh C., Diederich F.
Science,
2007
44.

Kirk K.L.
Organic Process Research and Development,
2008
45.

Soloshonok V., Aceña J., Sorochinsky A.
Synthesis,
2012
46.

Lombardo M., Easwar S., Pasi F., Trombini C., Dhavale D.D.
Tetrahedron,
2008