Abstract
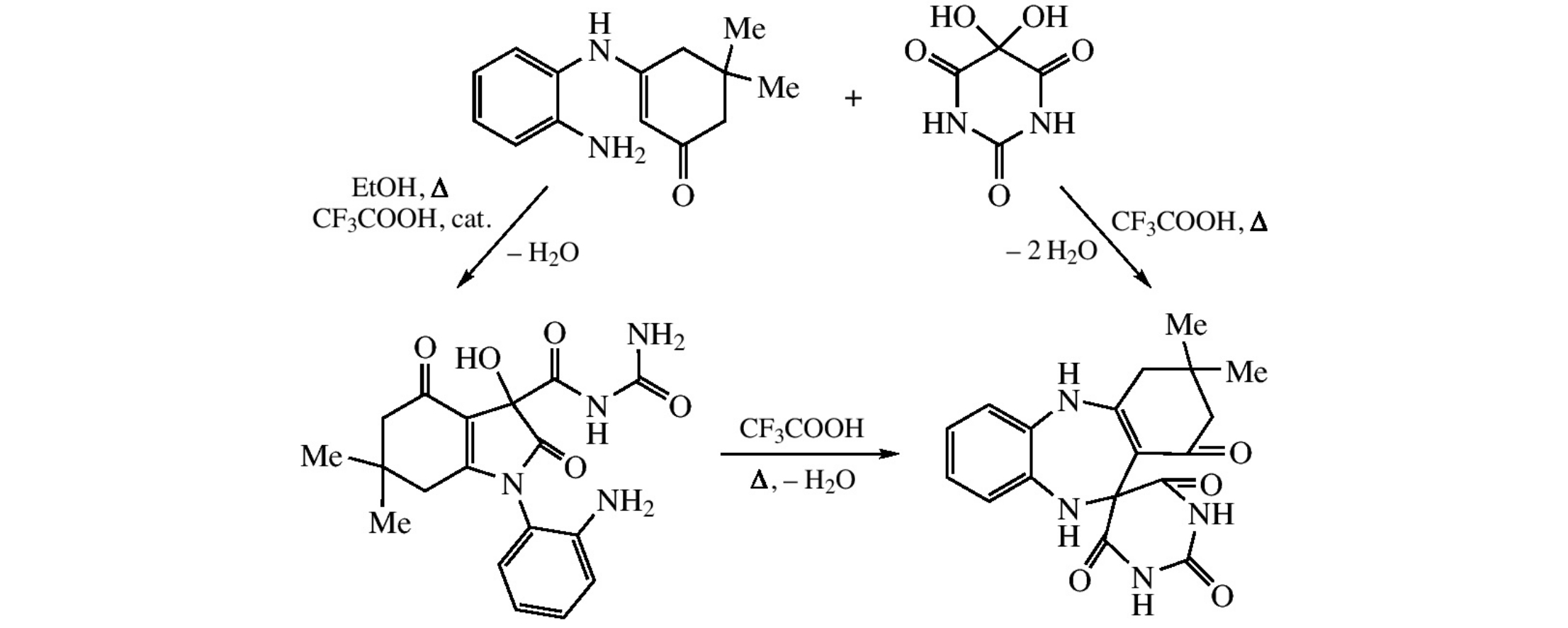
The reaction of 3-(2-aminophenylamino)-5,5-dimethyl-2-cyclohexen-1-one with alloxan results in 3,3-dimethyl-1,2,3,4,10,11-hexa- hydrospiro(1H-dibenzo[b,e][1,4]diazepine)-11,5’-pyrimidine-1,2’,4’,6’-tetraone or N-aminocarbonyl-1-(2-aminophenyl)-3-hydroxy- 6,6-dimethyl-2,4-dioxo-2,3,4,5,6,7-hexahydro-1H-indole-3-carboxamide. The latter undergoes isomerization on dissolution in DMSO or DMF to give 5-[2-(2-aminophenylamino)-4,4-dimethyl-6-oxocyclohexenyl]-5-hydroxypyrimidine-2,4,6(1H,3H,6H)-trione, thus demonstrating a new example of ring–ring tautomerism.
References
1.
![Synthesis of 3, 3-Dimethyl-2, 3, 4, 5, 10, 11-hexahydro-11-phenyl-1H-dibenzo [b, e] [1, 4] diazepin-1-one, a New Tricyclic System](/storage/images/resized/KBGMujzjwr0rGFxg8Kz0qvYdFHkcBo5w0GTtfiiU_small_thumb.webp)
MIYANO S., ABE N.
Chemical and Pharmaceutical Bulletin,
2011
2.
L. Yu. Ukhin, K. Yu. Suponitskii, E. N. Shepelenko, L. V. Belousova, Zh. I. Orlova and G. S. Borodkin, Russ. Chem. Bull., Int. Ed., 2011, 60, 1729.(Izv. Akad. Nauk, Ser. Khim., 2011, 1703).
3.
J.-F. Bonfanti, F.M. M. Doublet, O. Nyanguile, P.J.-M. B. Raboisson, A.-S. H. M. Rebstock and C. W. M. Boutton, WO Patent 2007026024A2, 2007 (Chem. Abstr., 2007, 146, 316950).
4.

Orlova Z.I., Ukhin L.Y., Suponitskii K.Y., Shepelenko E.N., Belousova L.V., Borodkin G.S., Popova O.S.
Russian Chemical Bulletin,
2013
5.
10.1016/j.mencom.2015.03.020_bib0025
Ukhin
Rostov-on-Don,
2012
6.
Ukhin L.Y., Orlova Z.I., Suponitsky K.Y., Shepelenko E.N., Belousova L.V.
Mendeleev Communications,
2014
7.
![Novel synthesis of oxonine derivatives from 3-[(2-aminophenyl)amino]-5,5-dimethyl-2-cyclohexene-1-one and o-quinones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ukhin L.Y., Suponitsky K.Y., Shepelenko E.N., Belousova L.V., Borodkin G.S.
Tetrahedron Letters,
2012
8.

Xue L., Jiang B., Tu M., Tu S.
Tetrahedron Letters,
2012
9.

Alekseyev V.V., Yakimovich S.I., Zerova I.V., Egorova M.B., Sinkkonen J.
Chemistry of Heterocyclic Compounds,
2014
10.
10.1016/j.mencom.2015.03.020_bib0050
Gordon
The Chemist's Companion: A Handbook of Practical Data, Techniques, and References,
1972