Abstract
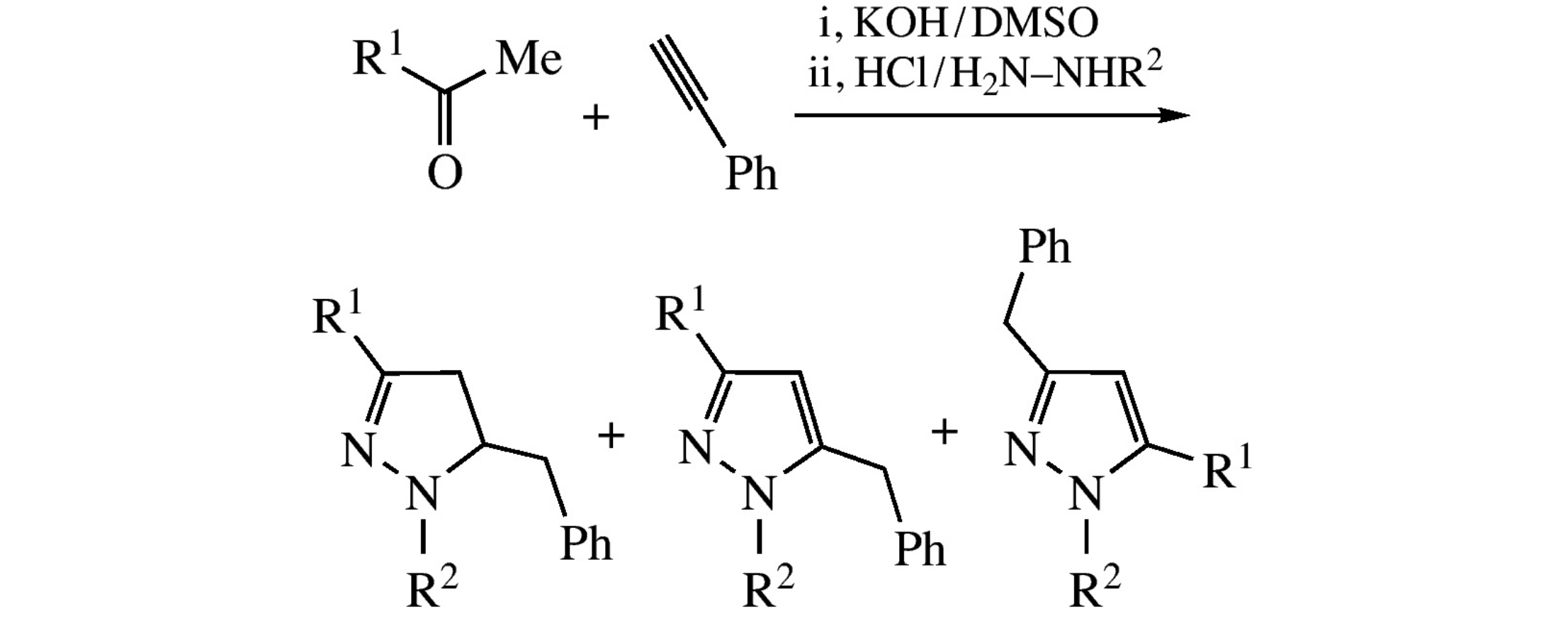
The sequential reaction of ketones with phenylacetylene in the presence of KOH/DMSO system followed by the treatment of the reaction mixture with HCl and monosubstituted hydrazines leads to pyrazolines and pyrazoles in up to 87% total yield.
References
1.

Trofimov B., Schmidt E., Ushakov I., Zorina N., Skital'tseva E., Protsuk N., Mikhaleva A.
Chemistry - A European Journal,
2010
2.

Trofimov B.A., Schmidt E.Y., Zorina N.V., Ivanova E.V., Ushakov I.A., Mikhaleva A.I.
Advanced Synthesis and Catalysis,
2012
3.

Trofimov B.A., Schmidt E.Y., Zorina N.V., Ivanova E.V., Ushakov I.A.
Journal of Organic Chemistry,
2012
4.
Schmidt E.Y., Zorina N.V., Tarasova O.A., Ushakov I.A., Trofimov B.A.
Mendeleev Communications,
2013
5.

Schmidt E.Y., Tatarinova I.V., Ivanova E.V., Zorina N.V., Ushakov I.A., Trofimov B.A.
Organic Letters,
2012
6.
Schmidt E.Y., Zorina N.V., Ivanova E.V., Tatarinova I.V., Ushakov I.A., Mikhaleva A.I., Trofimov B.A.
Mendeleev Communications,
2013
7.

Seijas J.A., Vazquez-Tato M.P., Castedo L., Estevez R.J., Ruiz M.
Journal of Organic Chemistry,
1992
8.

Wei X., Taylor R.J.
Chemical Communications,
1996
9.

Seijas J.A., Vázquez-Tato M.P., Entenza C., Montserrat Martínez M., Ònega M.G., Veiga S.
Tetrahedron Letters,
1998
11.
10.1016/j.mencom.2015.03.018_sbref0025b
Elkanzi
Int. J. Res. Pharm. Biomed. Sci.,
2013
12.

Katsiaouni S., Dechert S., Brückner C., Meyer F.
Chemical Communications,
2007
13.

Mayoral M.J., Ovejero P., Criado R., Cristina Lagunas M., Pintado-Alba A., Rosario Torres M., Cano M.
Journal of Organometallic Chemistry,
2011
14.

Cuerva C., Campo J.A., Ovejero P., Torres M.R., Cano M.
Dalton Transactions,
2014