Abstract
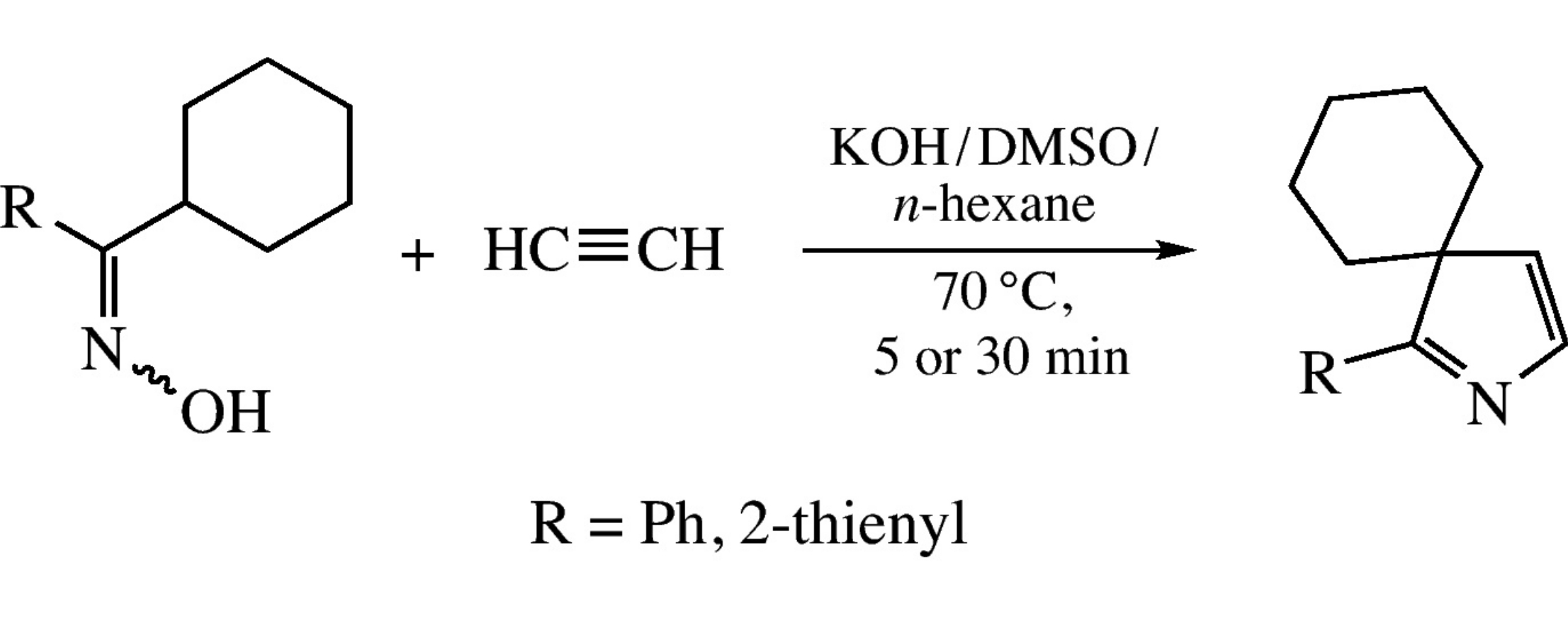
Cyclohexyl phenyl and cyclohexyl (2-thienyl) ketoximes react with acetylene under pressure in the two-phase KOH/DMSO/n-hexane system at 70°C to afford the corresponding spirocyclic 3H-pyrroles, 1-(het)aryl-2-azaspiro[4.5]deca-1,3-dienes, in 18–19% yields.
References
1.
M. P. Sammes and A. R. Katritzky, in Advances in Heterocyclic Chemistry, Academic Press, New York, 1982, vol. 32, p. 243.
2.

Sammes M.P.
Cumulative Index of Heterocyclic Systems,
1990
3.
B. A. Trofimov, S.G. Shevchenko, S.E. Korostova, A.I. Mikhaleva, M.V. Sigalov and L. B. Krivdin, Chem. Heterocycl. Compd., 1989, 25, 1314 (Khim. Geterotsikl. Soedin., 1989, 1566);
4.

Perrin S., Monnier K., Laude B.
Journal of Chemical Research,
1997
5.

Welch K.D., Smith P.L., Keller A.P., Myers W.H., Sabat M., Harman W.D.
Organometallics,
2006
6.
![Isomerization of 3H‐ to 2H‐[1]Benzothieno[3,2‐b]pyrroles and Synthesis of the First Merocyanine Dyes Based on Them](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Shimkin A.A., Shirinian V.Z., Nikalin D.M., Krayushkin M.M., Pivina T.S., Troitsky N.A., Vorontsova L.G., Starikova Z.A.
European Journal of Organic Chemistry,
2006
7.

Sobenina L.N., Tomilin D.N., Trofimov B.A.
Russian Chemical Reviews,
2014
8.
10.1016/j.mencom.2015.03.017_bib0015
Cirrincione
Farmaco,
1996
9.
10.1016/j.mencom.2015.03.017_sbref0020a
Cirrincione
Farmaco,
1992
10.

Padmavathi V., Radha Lakshmi T., Mahesh K., Padmaja A.
Chemical and Pharmaceutical Bulletin,
2009
11.

Hosaka K., Johnson A.P., Johnson A.W.
Tetrahedron Letters,
1978
12.

Lui K., Sammes M.P.
Journal of the Chemical Society Perkin Transactions 1,
1990
13.

Ichikawa J., Sakoda K., Mihara J., Ito N.
Journal of Fluorine Chemistry,
2006
14.
P. S. Silaichev, N.V. Kudrevatykh and A. N. Maslivets, Russ. J. Org. Chem., 2011, 47, 1602 (Zh. Org. Khim., 2011, 47, 1572);
15.
P. S. Silaichev, N.V. Kudrevatykh, P.A. Slepukhin and A. N. Maslivets, Russ. J. Org. Chem., 2013, 49, 860 (Zh. Org. Khim., 2013, 49, 876);
16.
P. S. Silaichev and A. N. Maslivets, Russ. J. Org. Chem., 2013, 49, 945 (Zh. Org. Khim., 2013, 49, 958).
17.
M. Yu. Belikov, O.V. Ershov, I.V. Lipovskaya, S.V. Fedoseev and O. E. Nasakin, Russ. J. Org. Chem., 2013, 49, 864.(Zh. Org. Khim., 2013, 49, 880).
18.

Trofimov B.A., Schmidt E.Y.
Russian Chemical Reviews,
2014
19.
R. J. Tedeschi, in Encyclopedia of Physical Science and Technology, 3rd edn., ed. R. A. Meyers, Academic Press, San Diego, 2001, vol. 1, p. 55;
20.
A. I. Mikhaleva and E. Yu. Schmidt, in Selected Methods for Synthesis and Modification of Heterocycles, ed. V. G. Kartsev, IBS Press, Moscow, 2002, vol. 1, p. 334;
21.
22.
Name Reactions in Heterocyclic Chemistry. II, ed. J. J. Li, Wiley- VCH, Hoboken, NJ,;1; 2011, p. 72;
23.
B. A. Trofimov, A.I. Mikhaleva, E. Yu. Schmidt and L. N. Sobenina, Khimiya pirrola. Novye stranitsy (Chemistry of Pyrrole. New Pages), Nauka, Novosibirsk, 2012 (in Russian).
24.
S. E. Korostova, S.G. Shevchenko and M. V. Sigalov, Chem. Heterocycl. Compd., 1991, 27, 1101.(Khim. Geterotsikl. Soedin., 1991, 1371).
25.
Shabalin D.A., Glotova T.E., Schmidt E.Y., Ushakov I.A., Mikhaleva A.I., Trofimov B.A.
Mendeleev Communications,
2014
26.
10.1016/j.mencom.2015.03.017_bib0070
Korostova
Zh. Org. Khim.,
1993