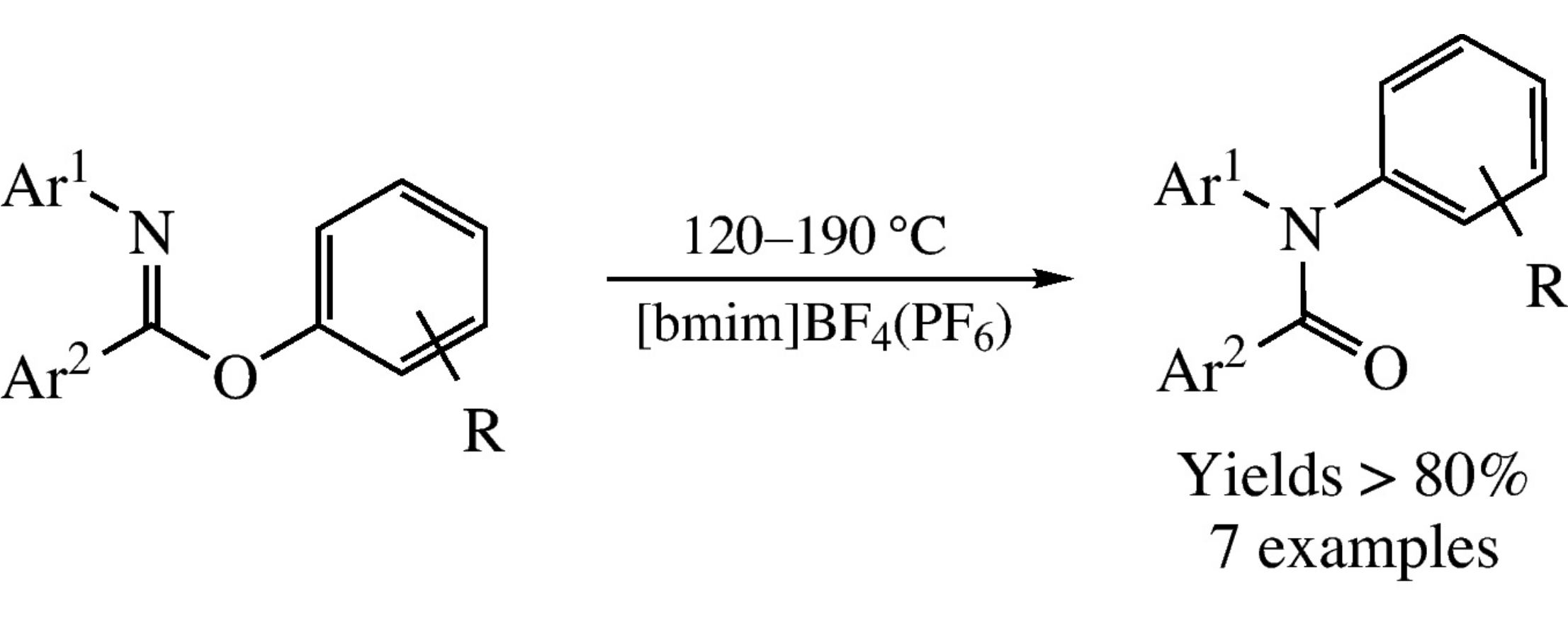
Abstract
The Chapman rearrangement of aryl N-arylbenzimidates to tertiary acyclic amides is accelerated in ionic liquids and proceeds at lower temperatures as 120–190°C.
References
1.

Wagner J., Wagner M.L., Hening W.A.
Annals of Pharmacotherapy,
1998
2.
![Synthetic analgesics. Synthesis and pharmacology of the diastereoisomers of N-[3-methyl-1-(2-phenylethyl)-4-piperidyl]-N-phenylpropanamide and N-[3-methyl-1-(1-methyl-2-phenylethyl)-4-piperidyl]-N-phenylpropanamide](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Van Bever W.F., Niemegeers C.J., Janssen P.A.
Journal of Medicinal Chemistry,
1974
3.
R. Anderson, T. Hokata, S.-F. Lee, R. Oey, T. Elich and S. Breazeale, US Patent 2008/02200461, 2008.
4.
10.1016/j.mencom.2015.03.016_sbref0010a
Barchless
J. Chem. Soc., Perkin Trans,
1987
5.

Thomas S.I., Falvey D.E.
Journal of Organic Chemistry,
2007
6.
J.-C. Kim, J. -Ch. Park, K.-S. Lee and J.-H. Jang, US Patent 2006/0258887 A1, 2006 (Chem. Abstr., 2006, 144, 146082);
7.
V. G. Babayan, J. Appl. Chem. USSR (Engl. Transl.), 1985, 58, 1100 (Zh. Prikl. Khim., 1985, 58, 1196);
8.
H. Kusaka, H. Takanashi, Yu. Ohgomori, K. Hakashima, G. Wen and T. Naito, Patent WO2006123473 (A1), 2006 (Chem. Abstr., 2006, 145, 505947).
9.

Hicks J.D., Hyde A.M., Cuezva A.M., Buchwald S.L.
Journal of the American Chemical Society,
2009
10.

Racine E., Monnier F., Vors J., Taillefer M.
Organic Letters,
2011
11.

Wang M., Yu H., You X., Wu J., Shang Z.
Chinese Journal of Chemistry,
2012
12.

Shang Z., Wu J., Wang M., Yu H.
Synthesis,
2013
13.

Mumm O., Hesse H., Volquartz H.
Berichte der deutschen chemischen Gesellschaft,
1915
14.

Chapman A.W.
Journal of the Chemical Society (Resumed),
1927
15.
J. W. Schulenberg and S. Archer, in Organic Reactions, ed. A. C. Cope, Wiley-VCH, New York, 1965, vol. 14, p. 1;
16.

Burdukovskii V.F., Mognonov D.M., Botoeva S.O., Mazurevskaya Z.P.
Russian Journal of Applied Chemistry,
2006
17.

Wiberg K.B., Rowland B.I.
Journal of the American Chemical Society,
1955
18.

Burdukovskii V.F., Mognonov D.M.
Russian Chemical Bulletin,
2008
19.

Wheeler O.H., Roman F., Santiago M.V., Quiles F.
Canadian Journal of Chemistry,
1969
20.

Welton T.
Chemical Reviews,
1999
21.
N. V. Plechkova and K. R. Seddon, in Methods and Reagents for Green Chemistry, eds. P. Tundo, A. Perosa and F. Zecchini, Wiley, New York, 2007, pp. 105-130;
22.

Plechkova N.V., Seddon K.R.
Chemical Society Reviews,
2008
23.

Ludwig R.
ChemSusChem,
2008
24.

Siddiqui I.R., Shireen, Shamim S., Waseem M.A., Abumhdi A.A., Srivastava A., Srivastava A.
Tetrahedron Letters,
2013
25.

Chakraborty B., Sharma C.D.
Tetrahedron Letters,
2013
26.

Shen Z., Goh K.K., Wong C.H., Loo W., Yang Y., Lu J., Loh T.
Chemical Communications,
2012
27.

Zlotin S.G., Makhova N.N.
Russian Chemical Reviews,
2010
28.
Zlotin S.G., Makhova N.N.
Mendeleev Communications,
2010
29.
N. V. Ignatyev, N.N. Makhova, A.S. Kulikov and M. A. Epishina, German Patent 102011014082 (A1), 2010;
30.
Epishina M.A., Kulikov A.S., Ignat’ev N.V., Schulte M., Makhova N.N.
Mendeleev Communications,
2011
31.

Petukhova V.Y., Fershtat L.L., Kachala V.V., Kuznetsov V.V., Khakimov D.V., Pivina T.S., Makhova N.N.
Journal of Heterocyclic Chemistry,
2013
32.
Pleshchev M.I., Petukhova V.Y., Kuznetsov V.V., Khakimov D.V., Pivina T.S., Struchkova M.I., Nelyubina Y.V., Makhova N.N.
Mendeleev Communications,
2013
33.
Pleshchev M.I., Epishina M.A., Kachala V.V., Kuznetsov V.V., Goloveshkin A.S., Bushmarinov I.S., Makhova N.N.
Mendeleev Communications,
2013
34.
Pleshchev M.I., Kachala V.V., Goloveshkin A.S., Bushmarinov I.S., Kuznetsov V.V., Khakimov D.V., Makhova N.N.
Mendeleev Communications,
2013
35.

Makhova N.N., Pleshchev M.I., Epishina M.A., Kulikov A.S.
Chemistry of Heterocyclic Compounds,
2014
36.
![Ionic liquid-promoted [3+2]-cycloaddition reactions of nitroformonitrile oxide generated by the cycloreversion of dinitrofuroxan](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Ovchinnikov I.V., Makhova N.N.
Tetrahedron Letters,
2014
37.

ULTRAMINE: A High-Capacity Polyethylene-Imine-Based Polymer and Its Application as a Scavenger Resin
Roice M., Christensen S.F., Meldal M.
Chemistry - A European Journal,
2004
38.

Schneider T.L., Halloran K.T., Hillner J.A., Conry R.R., Linton B.R.
Chemistry - A European Journal,
2013
39.
![Synthesis and Biological Evaluation of 2,3,5-Substituted [1,2,4]Thiadiazoles as Allosteric Modulators of Adenosine Receptors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
van den Nieuwendijk A.M., Pietra D., Heitman L., Göblyös A., IJzerman A.P.
Journal of Medicinal Chemistry,
2004
40.
![Synthesis, DNA-binding ability and anticancer activity of benzothiazole/benzoxazole–pyrrolo[2,1-c][1,4]benzodiazepine conjugates](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kamal A., Reddy K.S., Khan M.N., Shetti R.V., Ramaiah M.J., Pushpavalli S.N., Srinivas C., Pal-Bhadra M., Chourasia M., Sastry G.N., Juvekar A., Zingde S., Barkume M.
Bioorganic and Medicinal Chemistry,
2010
41.

Ghandi M., Hasani M., Salahi S.
Monatshefte fur Chemie,
2011
42.

Titus P.E., Forte P.A., Prokipcak J.M.
Canadian Journal of Chemistry,
1976
43.
10.1016/j.mencom.2015.03.016_bib0070
Chen
Asian J. Chem.,
2012