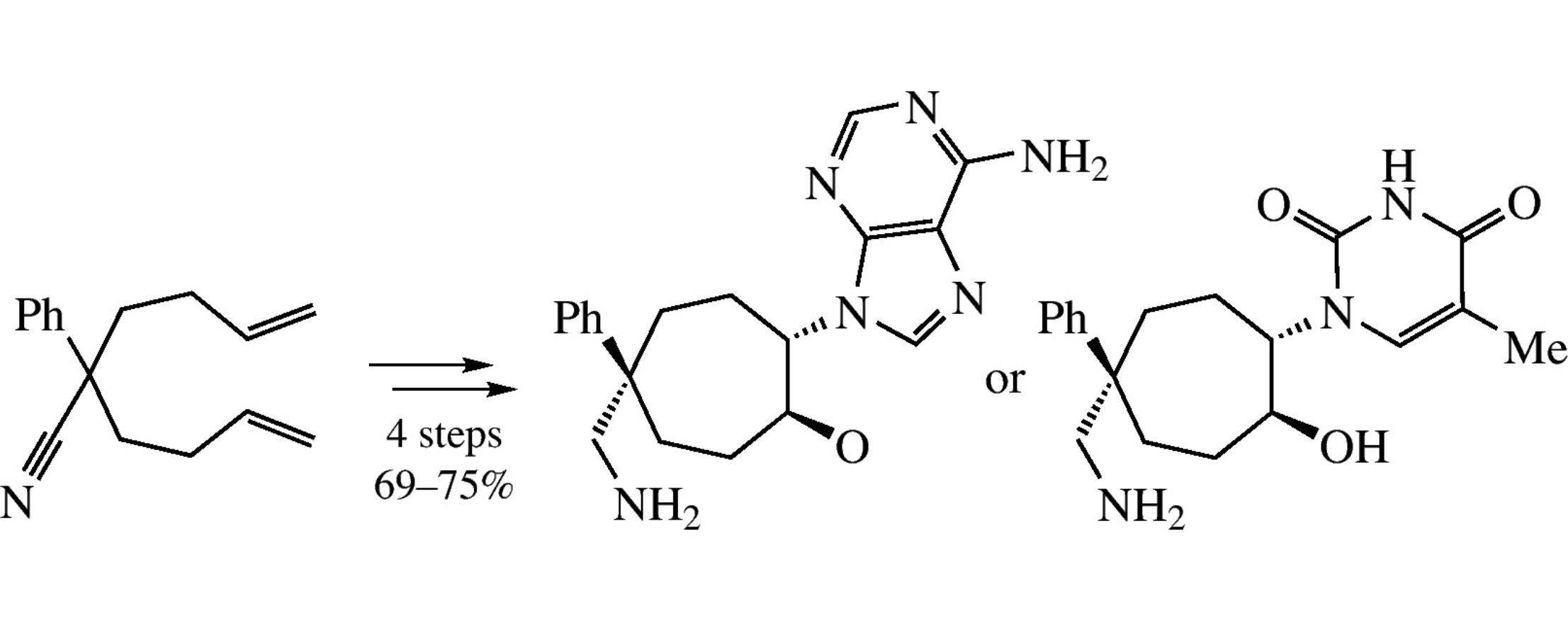
Abstract
Epoxidation of 1-phenylcyclohept-4-ene-1-carbonitrile gives mostly E-isomer of the desired epoxy derivative, whose ring opening with adenine or thymine proceeds stereoselectively providing access to novel cycloheptane nucleoside analogues.
References
1.

Dalençon S., Youcef R.A., Pipelier M., Maisonneuve V., Dubreuil D., Huet F., Legoupy S.
Journal of Organic Chemistry,
2011
2.

Zhang J., Chen Y., Huang Y., Jin H., Qiao R., Xing L., Zhang L., Yang Z., Zhang L.
Organic and Biomolecular Chemistry,
2012
3.

Gu P., Morral J., Wang J., Rozenski J., Busson R., Van Aerschot A., De Clercq E., Herdewijn P.
Antiviral Chemistry and Chemotherapy,
2003
4.

Viña D., Santana L., Uriarte E., Terán C.
Tetrahedron,
2005
5.

Blumberg P.M., Delclos K.B., Dunn J.A., Jaken S., Leach K.L., Yeh E.
Annals of the New York Academy of Sciences,
1983
6.

Nishizuka Y.
Nature,
1984
7.
10.1016/j.mencom.2015.03.014_bib0035
Singha
Arkivoc,
2003
8.

Ashworth I.W., Carboni D., Hillier I.H., Nelson D.J., Percy J.M., Rinaudo G., Vincent M.A.
Chemical Communications,
2010
9.
Y.-J. Wu and H. He, US Patent 2006/19992 A1, 2006.
10.
R. J. Bull, E.A. Skidmore, R.L. Ford, A.N. Mather and A. Mete, US Patent 2011/172237 A1, 2011.
11.

Rishel M.J., Thomas C.J., Tao Z., Vialas C., Leitheiser C.J., Hecht S.M.
Journal of the American Chemical Society,
2003
12.

Marco-Contelles J., de Opazo E.
Journal of Organic Chemistry,
2000
13.
Liboska R., Masojídková M., Rosenberg I.
Collection of Czechoslovak Chemical Communications,
2002
14.
Liboska R., Masojídková M., Rosenberg I.
Collection of Czechoslovak Chemical Communications,
2002
15.

Denmark S.E., Forbes D.C., Hays D.S., DePue J.S., Wilde R.G.
Journal of Organic Chemistry,
1995
16.

Lane B.S., Burgess K.
Chemical Reviews,
2003
17.

Zhang L., Peritz A., Meggers E.
Journal of the American Chemical Society,
2005
18.

Yang S., Murray W.V.
Tetrahedron Letters,
2008
19.

Jordheim L.P., Durantel D., Zoulim F., Dumontet C.
Nature Reviews Drug Discovery,
2013
20.

Fülöp F., Huber I., Bernáth G., Hönig H., Seufer-Wasserthal P.
Synthesis,
1991
21.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
22.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007