Abstract
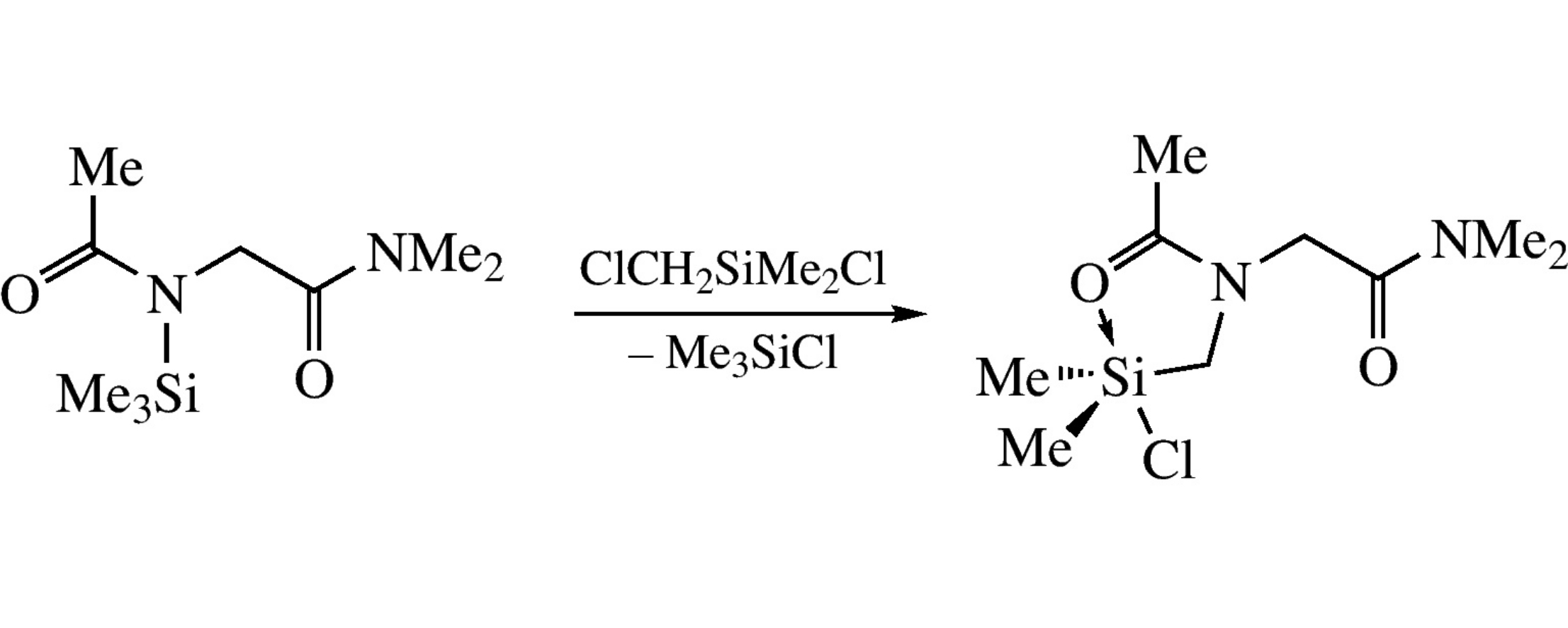
Regioselective reaction of N-trimethylsilyl-N-acetylglycine N’,N’-dimethylamide with chloro(chloromethyl)dimethylsilane yields chlorosilane MeC(O)N(CH2SiMe2Cl)CH2C(O)NMe2 with a five-membered C,O-chelate ring involving the N-acetyl group rather than the six-membered ring involving the N’,N’-dimethylamide fragment. According to X-ray data, the pentacoordinate silicon atom in the product has a TBP environment with the halogen and oxygen atoms in axial positions.
References
1.
(a) D. Kost and I. Kalikhman, in The Chemistry of Organic Silicon Compounds, eds. Z. Rappoport and Y. Apeloig,;1; J. Willey, Chichester, 1998, vol. 2, part 1, p. 1339; (b) M. G. Voronkov, V.A. Pestunovich and Yu. I. Baukov, Organomet. Chem. USSR,;1; 1991, 4, 593 (Metalloorg. Khim., 1991, 4, 1210); (c) C. Chuit, R.J. P. Corriu, C. Reyé and J. C. Young, Chem. Rev., 1993, 93, 1371.
2.

Macharashvili A.A., Shklover V.E., Struchkov Y.T., Oleneva G.I., Kramarova E.P., Shipov A.G., Baukov Y.I.
Journal of the Chemical Society Chemical Communications,
1988
3.
10.1016/j.mencom.2015.03.011_sbref0010b
Baukov Yu.
Zh. Obshch. Khim.,
1989
4.
10.1016/j.mencom.2015.03.011_sbref0010c
Artamkina
Zh. Obshch. Khim.,
1993
5.
10.1016/j.mencom.2015.03.011_sbref0010d
Artamkina
Zh. Obshch. Khim.,
1994
6.

Shipov A.G., Kramarova E.P., Murasheva T.P., Korlyukov A.A., Pogozhikh S.A., Tarasenko S.A., Negrebetskii V.V., Yakovlev I.P., Baukov Y.I.
Russian Journal of General Chemistry,
2011
7.

Negrebetsky V.V., Tandura S.N., Baukov Y.I.
Russian Chemical Reviews,
2009
8.

Negrebetsky V.V., Baukov Y.I.
Russian Chemical Bulletin,
1997
9.

Negrebetsky V.V., Taylor P.G., Kramarova E.P., Bylikin S.Y., Belavin I.Y., Shipov A.G., Bassindale A.R., Baukov Y.I.
Journal of Organometallic Chemistry,
2006
10.

Sidorkin V.F., Belogolova E.F., Pestunovich V.A.
Journal of Molecular Structure THEOCHEM,
2001
11.

Sidorkin V.F., Vladimirov V.V., Voronkov M.G., Pestunovich V.A.
Journal of Molecular Structure THEOCHEM,
1991
12.

Bassindale A.R., Parker D.J., Taylor P.G., Auner N., Herrschaft B.
Journal of Organometallic Chemistry,
2003
13.

Ovchinnikov Y.E., Macharashvili A.A., Struchkov Y.T., Shipov A.G., Baukov Y.I.
Journal of Structural Chemistry,
1994
14.

Bassindale A.R., Borbaruah M., Glynn S.J., Parker D.J., Taylor P.G.
Journal of the Chemical Society Perkin Transactions 2,
1999
15.

Bassindale A.
Journal of Organometallic Chemistry,
2001
16.
(a) A. G. Shipov, E.P. Kramarova, O.B. Artamkina, G.I. Oleneva and Yu. I. Baukov, J. Gen. Chem. USSR, 1991, 61, 1770 (Zh. Obshch. Khim., 1991, 61, 1914); (b) M. Sohail, A.R. Bassindale, P.G. Taylor, A.A. Korlyukov, D.E. Arkhipov, L. Male, S.J. Coles and M. B. Hursthouse, Organometallics, 2013, 32, 1721; (c) A. G. Shipov, E.P. Kramarova, H.C. Fang, D.E. Arkhipov, A.A. Nikolin, S. Yu. Bylikin, V.V. Negrebetsky, A.A. Korlyukov, N.A. Voronina, A.R. Bassindale, P.G. Taylor and Yu. I. Baukov, J. Organomet. Chem., 2013, 741-742, 114; (d) A. A. Nikolin, D.E. Arkhipov, A.G. Shipov, E.P. Kramarova, N.A. Koval’chuk, A.A. Korlyukov, V.V. Negrebetsky, Yu. I. Baukov, A.R. Bassindale, P.G. Taylor, A.A. Bowden and S. Yu. Bylikin, Chem. Heterocycl. Compd., 2011, 47, 1565.(Khim. Geterotsikl. Soedin., 2011, 1869).
17.

Pestunovich V.A., Kirpichenko S.V., Lazareva N.F., Albanov A.I., Voronkov M.G.
Journal of Organometallic Chemistry,
2007
18.

Allen F.H., Kennard O., Watson D.G., Brammer L., Orpen A.G., Taylor R.
Journal of the Chemical Society Perkin Transactions 2,
1987
19.

Belogolova E.F., Sidorkin V.F.
Journal of Physical Chemistry A,
2013
20.

Mazurkiewicz R.
Synthesis,
1992
21.
M. J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, Ö. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision D.01, Gaussian, Inc., Wallingford CT, 2009.