Abstract
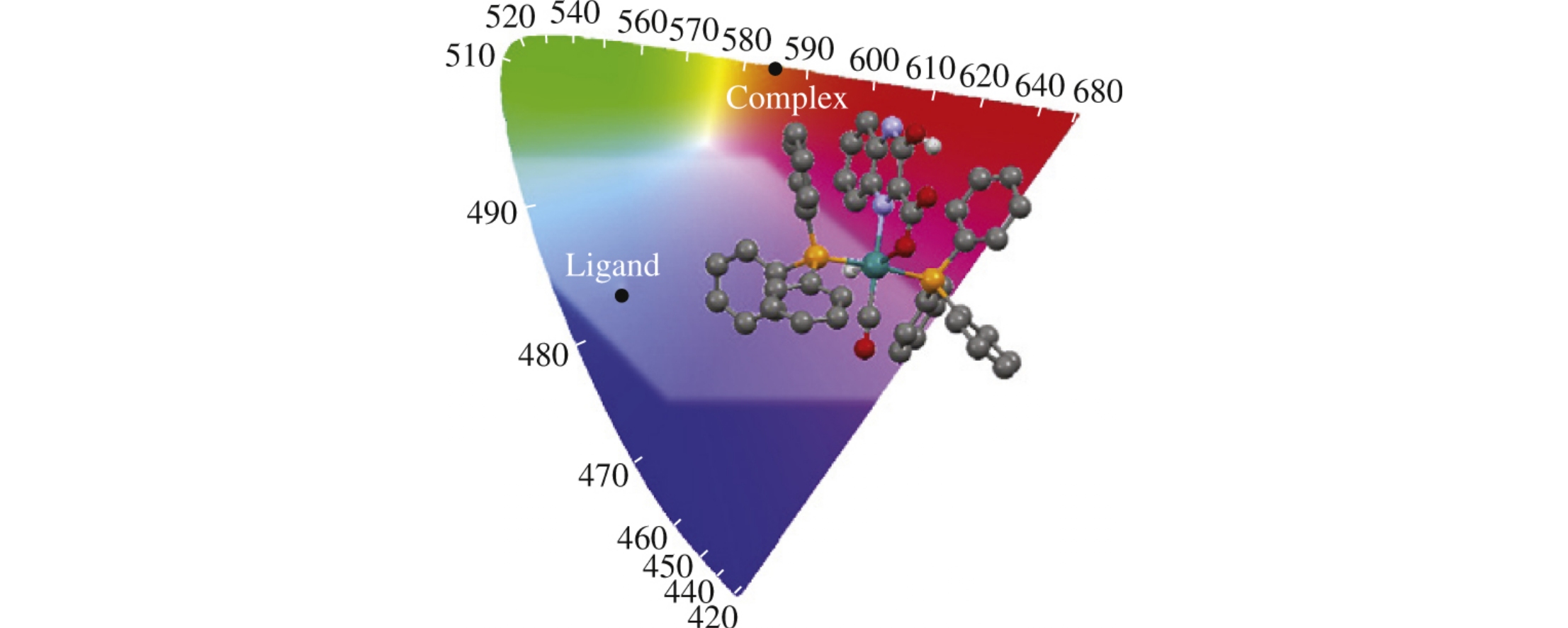
The [RuH(CO)(hqxc)(PPh3)2] complex, where hqxc is 3-hydroxy-2-quinoxalinecarboxylate, exhibits strong phosphorescence (quantum yield F = 0.0078) in the solid state giving rise to a significantly Stokes-shifted spectrum (excitation at 484nm and emission at 687nm); the lifetime of 6.5μs is very long compared with 0.68ns measured for the free hqxc ligand.
References
1.

Perruchas S., Le Goff X.F., Maron S., Maurin I., Guillen F., Garcia A., Gacoin T., Boilot J.
Journal of the American Chemical Society,
2010
2.

Armaroli N., Accorsi G., Holler M., Moudam O., Nierengarten J.-., Zhou Z., Wegh R. ., Welter R.
Advanced Materials,
2006
3.

Zhang Q., Zhou Q., Cheng Y., Wang L., Ma D., Jing X., Wang F.
Advanced Materials,
2004
4.

Nishikawa M., Nomoto K., Kume S., Inoue K., Sakai M., Fujii M., Nishihara H.
Journal of the American Chemical Society,
2010
5.

Chen J., Wu B., Gu W., Cao X., Wen H., Hong R., Liao J., Su B.
Transition Metal Chemistry,
2011
6.
Karasik A.A., Balueva A.S., Musina E.I., Sinyashin O.G.
Mendeleev Communications,
2013
7.
Solodukhin N.N., Utochnikova V.V., Lepnev L.S., Kuzmina N.P.
Mendeleev Communications,
2014
8.
Malecki J.G., Maron´ A., Gryca I., Serda M.
Mendeleev Communications,
2014
9.

Małecki J.G., Maroń A.
Transition Metal Chemistry,
2012
10.

Małecki J.G., Maroń A.
Transition Metal Chemistry,
2012
11.

12.
![Sensitized Near-Infrared Luminescence From NdIII, YbIII and ErIII Complexes by Energy-Transfer From Ruthenium 1,3-Bis([1,10]Phenanthroline-[5,6-d]-Imidazol-2 -yl)Benzene](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Singaravadivel S., Velayudham M., Babu E., Mareeswaran P.M., Lu K., Rajagopal S.
Journal of Fluorescence,
2013
13.

Hammes-Schiffer S.
Accounts of Chemical Research,
2001
14.

Nagaoka S., Nakamura A., Nagashima U.
Journal of Photochemistry and Photobiology A: Chemistry,
2002
15.

Klymchenko A.S., Pivovarenko V.G., Ozturk T., Demchenko A.P.
New Journal of Chemistry,
2003
16.

Park S., Kwon O., Kim S., Park S., Choi M., Cha M., Park S.Y., Jang D.
Journal of the American Chemical Society,
2005
17.

Lim S., Seo J., Park S.Y.
Journal of the American Chemical Society,
2006
18.
![Extensive spectral tuning of the proton transfer emission from 550 to 675 nm via a rational derivatization of 10-hydroxybenzo[h]quinoline.](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Chen K., Hsieh C., Cheng Y., Lai C., Chou P.
Chemical Communications,
2006
19.

Klymchenko A.S., Yushchenko D.A., Mély Y.
Journal of Photochemistry and Photobiology A: Chemistry,
2007
20.

Badoğlu S., Yurdakul Ş.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2013
21.
10.1016/j.mencom.2015.03.007_bib0105
Sheldrick
2008