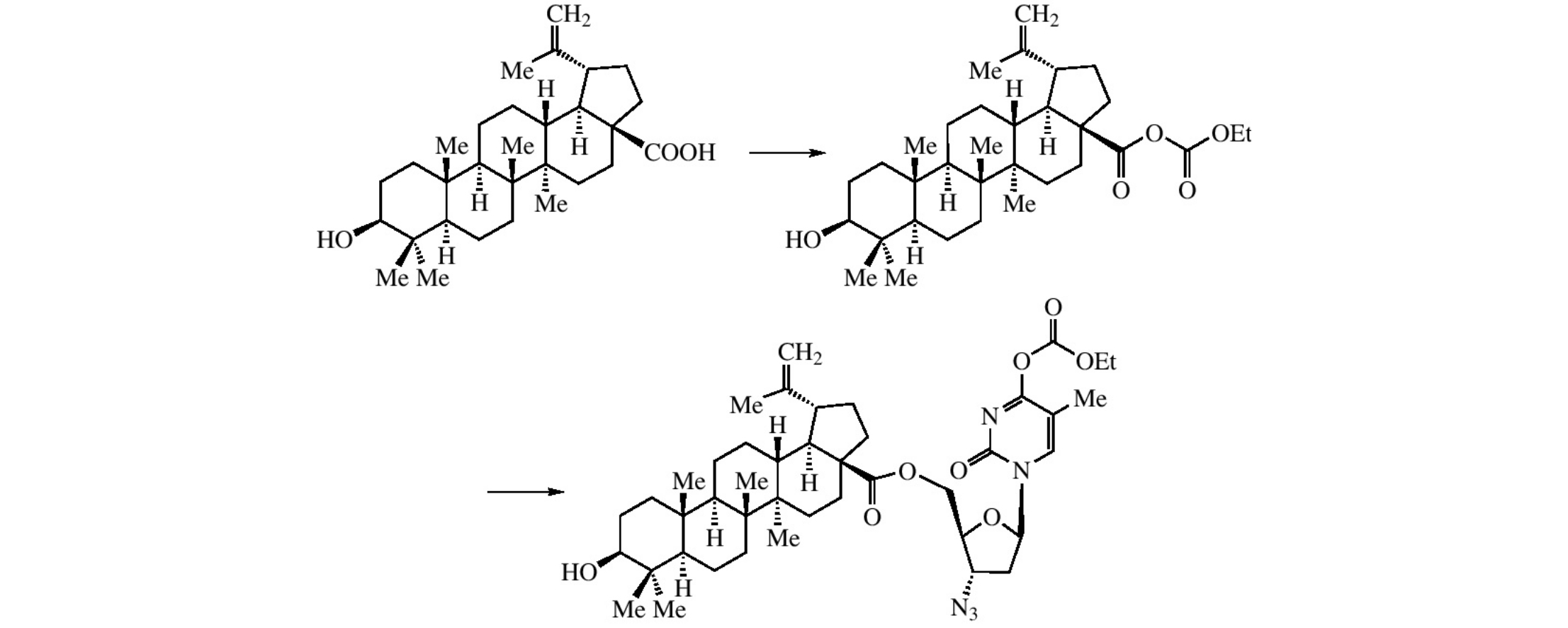
Abstract
One-pot synthesis of ester-linked conjugates of betulinic, ursolic or oleanolic acids with anti-HIV drugs AZT, derivatives of AZT and 3TC was performed, the resulting conjugates having shown high anti-HIV activities.
References
1.

Fujioka T., Kashiwada Y., Kilkuskie R.E., Cosentino L.M., Ballas L.M., Jiang J.B., Janzen W.P., Chen I., Lee K.
Journal of Natural Products,
1994
2.

Pengsuparp T., Cai L., Fong H.H., Kinghorn A.D., Pezzuto J.M., Wani M.C., Wall M.E.
Journal of Natural Products,
1994
3.
Govdi A.I., Vasilevsky S.F., Sokolova N.V., Sorokina I.V., Tolstikova T.G., Nenajdenko V.G.
Mendeleev Communications,
2013
4.

Yogeeswari P., Sriram D.
Current Medicinal Chemistry,
2005
5.

Kashiwada Y., Hashimoto F., Cosentino L.M., Chen C., Garrett P.E., Lee K.
Journal of Medicinal Chemistry,
1996
6.
![[d4U]-butyne-[HI-236] as a non-cleavable, bifunctional NRTI/NNRTI HIV-1 reverse-transcriptase inhibitor](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Hunter R., Muhanji C.I., Hale I., Bailey C.M., Basavapathruni A., Anderson K.S.
Bioorganic and Medicinal Chemistry Letters,
2007
7.

Chen S., Wang Y., Jin Y., Tian X., Zheng Y., Luo D., Tu Y.
Bioorganic and Medicinal Chemistry Letters,
2007
8.

Matsumoto H., Matsuda T., Nakata S., Mitoguchi T., Kimura T., Hayashi Y., Kiso Y.
Bioorganic and Medicinal Chemistry,
2001
9.
![Potential multifunctional inhibitors of HIV-1 reverse transcriptase. Novel [AZT]-[TSAO-T] and [d4T]-[TSAO-T] heterodimers modified in the linker and in the dideoxynucleoside region](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Velázquez S., Tuñón V., Jimeno M.L., Chamorro C., De Clercq E., Balzarini J., Camarasa M.J.
Journal of Medicinal Chemistry,
1999
10.

Matsumoto H., Kimura T., Hamawaki T., Kumagai A., Goto T., Sano K., Hayashi Y., Kiso Y.
Bioorganic and Medicinal Chemistry,
2001
11.

Matsumoto H., Hamawaki T., Ota H., Kimura T., Goto T., Sano K., Hayashi Y., Kiso Y.
Bioorganic and Medicinal Chemistry Letters,
2000
12.

Xiong J., Kashiwada Y., Chen C., Qian K., Morris-Natschke S.L., Lee K., Takaishi Y.
Bioorganic and Medicinal Chemistry,
2010
13.

Bori I.D., Hung H., Qian K., Chen C., Morris-Natschke S.L., Lee K.
Tetrahedron Letters,
2012
14.

Dang Thi T.A., Kim Tuyet N.T., Pham The C., Thanh Nguyen H., Ba Thi C., Doan Duy T., D’hooghe M., Van Nguyen T.
Bioorganic and Medicinal Chemistry Letters,
2014
15.
10.1016/j.mencom.2015.03.004_sbref0060b
Tuyet Anh
Tetrahedron Lett,
2014