Abstract
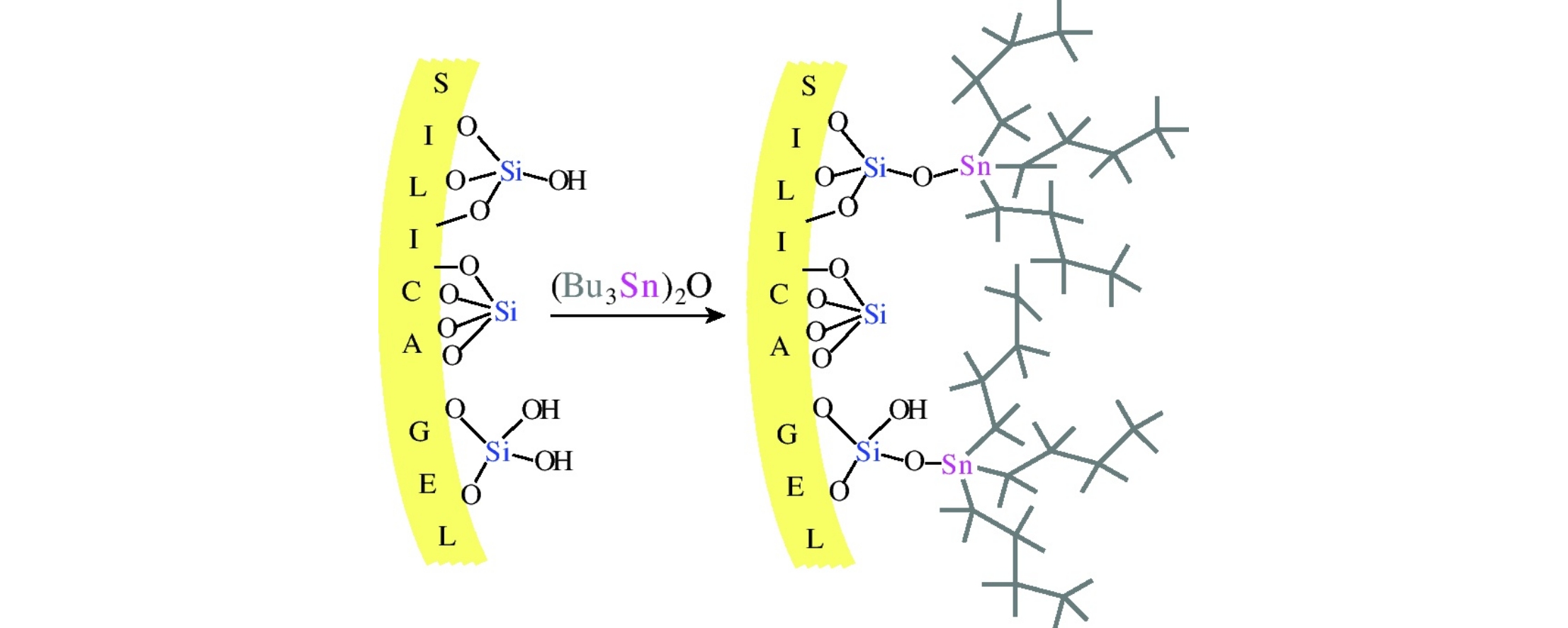
Bis(tri-n-butyltin) oxide quickly reacts with chromatographic silica gel even when a solution of this oxide is filtered through silica gel, which results in stannylation of a fraction of the surface hydroxyl groups of silica gel to give n-Bu3SnOSi° moieties. The calculated surface density of these moieties, 0.85 Sn groups per nm2, corresponds to complete coverage of silica gel surface with n-Bu groups.
References
1.

Fouquet E.
Chemistry of Functional Groups,
2002
2.

Carland M.W., Schiesser C.H.
Chemistry of Functional Groups,
2002
3.

Lukevics E., Pudova O.
Chemistry of Functional Groups,
2002
4.

Duffy J.A., Harston P., Wardell J.L., Smith P.J.
Applied Organometallic Chemistry,
1990
5.

Ballivet-Tkatchenko D., dos Santos J.H., Malisova M.
Langmuir,
1993
6.

Nedez C., Theolier A., Lefebvre F., Choplin A., Basset J.M., Joly J.F.
Journal of the American Chemical Society,
1993
7.

Nedez C., Choplin A., Lefebvre F., Basset J.
Inorganic Chemistry,
1994
8.

Nedez C., Lefebvre F., Choplin A., Niccolai G.P., Basset J., Benazzi E.
Journal of the American Chemical Society,
1994
10.

Millot N., Santini C.C., Lefebvre F., Basset J.
Comptes Rendus Chimie,
2004
11.
Modern Surface Organometallic Chemistry, eds. J.-M. Basset, R. Psaro, D. Roberto and R. Ugo, Wiley-VCH, Weinheim, 2009, pp. 31, 168, 426.
12.
Lapitskaya M.A., Vasiljeva L.L., Pivnitsky K.K.
Mendeleev Communications,
2013
13.

Bartram M.E., Michalske T.A., Rogers J.W.
The Journal of Physical Chemistry,
1991
14.
L. T. Zhuravlev, in Colloidal Silica. Fundamental and Applications, eds. H. E. Bergna and W. O. Roberts, Taylor & Francis Group, Boca Raton, 2006, p. 262.
15.

Crich D., Sun S.
Journal of Organic Chemistry,
1996
16.

Jousseaume B., Chanson E., Pereyre M.
Organometallics,
1986
17.

18.

Curthoys G., Davydov V.Y., Kiselev A.V., Kiselev S.A., Kuznetsov B.V.
Journal of Colloid and Interface Science,
1974
19.

Fyfe C.A., Gobbi G.C., Kennedy G.J.
The Journal of Physical Chemistry,
1985
20.

Leonardelli S., Facchini L., Fretigny C., Tougne P., Legrand A.P.
Journal of the American Chemical Society,
1992
21.

Adachi M., Lefebvre F., Basset J.
Chemistry Letters,
1996
22.

Basset J., Lefebvre F., Santini C.
Coordination Chemistry Reviews,
1998
23.

Maciel G.E., Sindorf D.W.
Journal of the American Chemical Society,
1980
24.

Sindorf D.W., Maciel G.E.
The Journal of Physical Chemistry,
1982
25.

Tanimura H., Sekine M., Hata T.
Tetrahedron,
1986
26.

Harpp D.N., Gingras M.
Journal of the American Chemical Society,
1988
27.

Gingras M., Chan T.H.
Tetrahedron Letters,
1989
28.

Kamiyama T., Inoue M., Enomoto S.
Chemistry Letters,
1989
29.

Kamiyama T., Inoue M., Kashiwagi H., Enomoto S.
Bulletin of the Chemical Society of Japan,
1990
30.

Salomon C.J., Mata E.G., Mascaretti O.A.
Tetrahedron Letters,
1991
31.

Salomon C.J., Mata E.G., Mascaretti O.A.
Journal of Organic Chemistry,
1994
32.

Isac-García J., Calvo-Flores F., Hernández-Mateo F., Santoyo-González F.
European Journal of Organic Chemistry,
2001
33.

34.

Zhang C.A., Erhardt P.W.
Synthetic Communications,
2011
35.

Harrowven D.C., Curran D.P., Kostiuk S.L., Wallis-Guy I.L., Whiting S., Stenning K.J., Tang B., Packard E., Nanson L.
Chemical Communications,
2010
36.

Kachala V.V., Khemchyan L.L., Kashin A.S., Orlov N.V., Grachev A.A., Zalesskiy S.S., Ananikov V.P.
Russian Chemical Reviews,
2013