Abstract
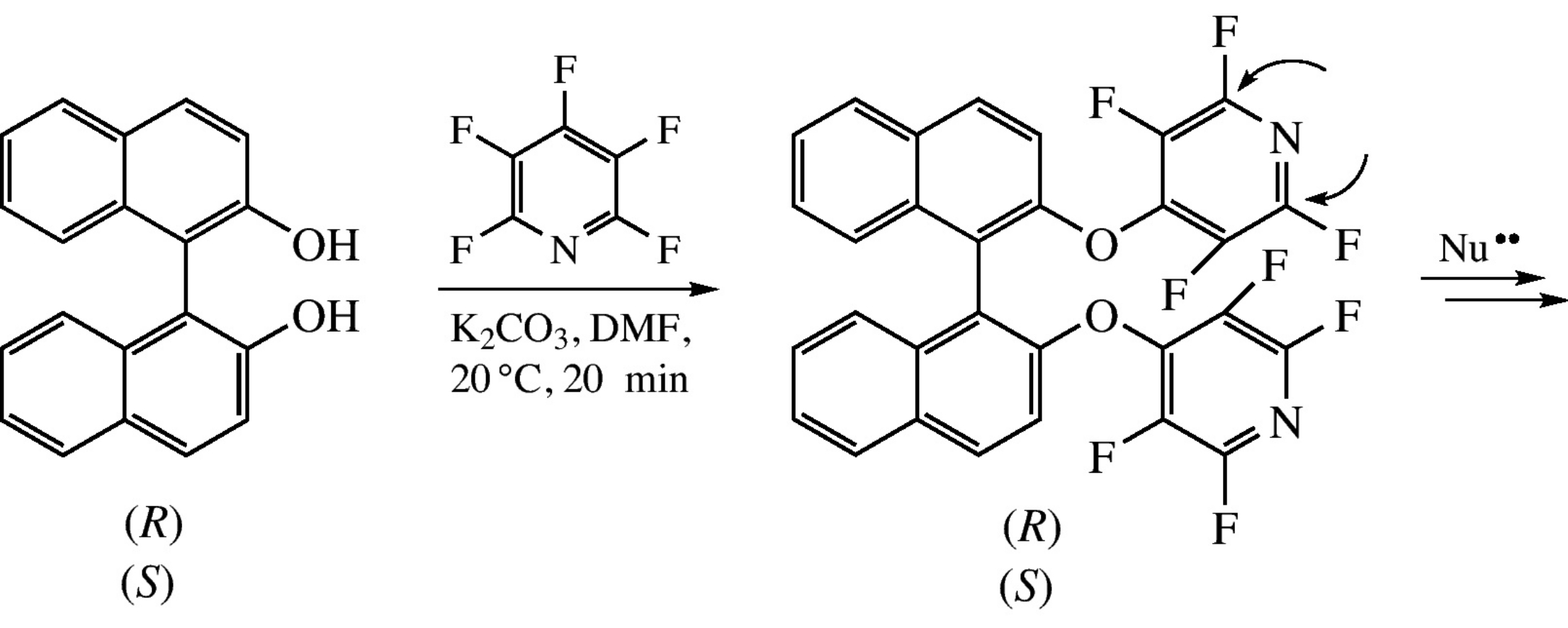
(R)- and (S)-binaphthyl-2,2’-diols (BINOLs) smoothly react with pentafluoropyridine without racemization to give (R)- and (S)-2,2’- bis(tetrafluoropyridin-4-yloxy)-1,1’-binaphthyls. The 2(6)-positioned pyridine fluorine atoms can be replaced with amino moieties by reactions with amines.
References
1.

Schenker S., Zamfir A., Freund M., Tsogoeva S.B.
European Journal of Organic Chemistry,
2011
2.

3.

Chen Y., Yekta S., Yudin A.K.
Chemical Reviews,
2003
4.

Kočovský P., Vyskočil Š., Smrčina M.
Chemical Reviews,
2003
6.
Koltunov K.Y., Chernov A.N., Ostashevskaya L.A., Gribov P.A.
Mendeleev Communications,
2014
7.

Dabbagh H.A., Najafi-Chermahini A., Banibairami S.
Tetrahedron Letters,
2006
8.
![Synthesis of BINOL-containing oxacalix[4]arenes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Capici C., Gattuso G., Notti A., Parisi M.F., Bruno G., Nicolò F., Pappalardo S.
Tetrahedron Letters,
2011
9.

Gandhi P., Huang B., Gallucci J.C., Parquette J.R.
Organic Letters,
2001
10.

Qu X., Li T., Zhu Y., Sun P., Yang H., Mao J.
Organic and Biomolecular Chemistry,
2011
11.

Kyba E.P., Gokel G.W., De Jong F., Koga K., Sousa L.R., Siegel M.G., Kaplan L., Sogah G.D., Cram D.J.
Journal of Organic Chemistry,
1977
12.

Chambers R.D., Hoskin P.R., Sandford G., Yufit D.S., Howard J.A.
Journal of the Chemical Society Perkin Transactions 1,
2001
13.

Chambers R.D.
Journal of Fluorine Chemistry,
2010
14.

Litvak V.V., Mainagashev I.Y., Bukhanets O.G.
Russian Chemical Bulletin,
2007
15.

Vlasov V.M., Aksenov V.V., Rodionov P.P., Beregovaya I.V., Shchegoleva L.N.
Russian Journal of Organic Chemistry,
2002
16.

Jarman M., McCague R.
Journal of the Chemical Society Chemical Communications,
1984