Abstract
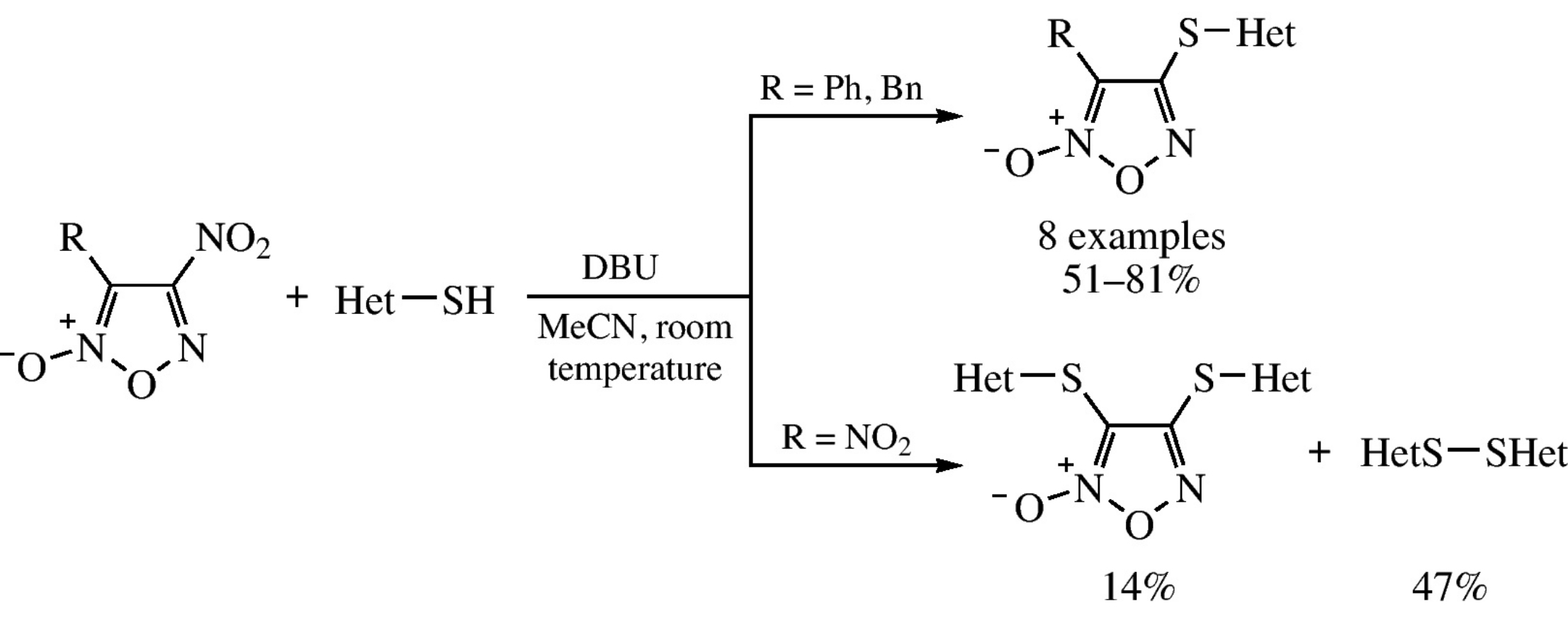
Hetarylthiofuroxans were synthesized by nucleophilic substitution of the nitro group in 4-nitrofuroxans under the action of hetarylthiols in the DBU–MeCN system at room temperature, reactivity of 4-nitrofuroxans being dependent on the C(3)-substituent.
References
1.
![Vasorelaxant and antiplatelet activity of 4,7-dimethyl-1,2,5-oxadiazolo[3,4-d]pyridazine 1,5,6-trioxide: Role of soluble guanylate cyclase, nitric oxide and thiols](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kots A.Y., Grafov M.A., Khropov Y.V., Betin V.L., Belushkina N.N., Busygina O.G., Yazykova M.Y., Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Medvedeva N.A., Bulargina T.V., Severina I.S.
British Journal of Pharmacology,
2000
2.

Cena C., Lolli M.L., Lazzarato L., Guaita E., Morini G., Coruzzi G., McElroy S.P., Megson I.L., Fruttero R., Gasco A.
Journal of Medicinal Chemistry,
2003
3.

Buonsanti M.F., Bertinaria M., Di Stilo A., Cena C., Fruttero R., Gasco A.
Journal of Medicinal Chemistry,
2007
4.

Boiani L., Aguirre G., González M., Cerecetto H., Chidichimo A., Cazzulo J.J., Bertinaria M., Guglielmo S.
Bioorganic and Medicinal Chemistry,
2008
5.

Chegaev K., Cena C., Giorgis M., Rolando B., Tosco P., Bertinaria M., Fruttero R., Carrupt P., Gasco A.
Journal of Medicinal Chemistry,
2008
6.

Schiefer I.T., VandeVrede L., Fa’ M., Arancio O., Thatcher G.R.
Journal of Medicinal Chemistry,
2012
7.

Ferioli R., Fazzini A., Folco G.C., Fruttero R., Calvino R., Gasco A., Bongrani S., Civelli M.
Pharmacological Research,
1993
8.

Lu Y., Kandel E.R., Hawkins R.D.
Journal of Neuroscience,
1999
9.

Bon C.L., Garthwaite J.
Journal of Neuroscience,
2003
10.

Haley J.E., Wilcox G.L., Chapman P.F.
Neuron,
1992
11.

Thatcher G.R., Nicolescu A.C., Bennett B.M., Toader V.
Free Radical Biology and Medicine,
2004
12.

Ferioli R., Folco G.C., Ferretti C., Gasco A.M., Medana C., Fruttero R., Civelli M., Gasco A.
British Journal of Pharmacology,
1995
13.

Fershtat L.L., Struchkova M.I., Goloveshkin A.S., Bushmarinov I.S., Makhova N.N.
Heteroatom Chemistry,
2014
14.
Ovchinnikov I.V., Makhova N.N., Khmel’nitskii L.I.
Mendeleev Communications,
1993
15.

Ovchinnikov I.V., Makhova N.N., Khmel'nitskii L.I.
Russian Chemical Bulletin,
1995
16.

Sharma R., Pandey A.K., Shivahare R., Srivastava K., Gupta S., Chauhan P.M.
Bioorganic and Medicinal Chemistry Letters,
2014
17.

Mohebbi S., Falcón-Pérez J.M., González E., Millet O., Mato J.M., Kobarfard F.
Chemical and Pharmaceutical Bulletin,
2012
18.
![Thiourea and thiosemicarbazide derivatives: Structure, transformations, and pharmacological activity. Part II. Antihypoxic activity of 1,2,4-triazino[5,6-b]indole derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Tomchin A.B., Uryupov O.Y., Zhukova T.I., Kuznetsova T.A., Kostycheva M.V., Smirnov A.V.
Pharmaceutical Chemistry Journal,
1997
19.

Zhao Y., Chen Z., Chen Y., Xu J., Li J., Jiang X.
Journal of the American Chemical Society,
2013
20.

Laura Almajan G., Innocenti A., Puccetti L., Manole G., Barbuceanu S., Saramet I., Scozzafava A., Supuran C.T.
Bioorganic and Medicinal Chemistry Letters,
2005
21.
10.1016/j.mencom.2015.01.013_bib0105
Terrier
Modern Nucleophilic Aromatic Substitution,
2013
22.

Makhova N.N., Kulikov A.S.
Russian Chemical Reviews,
2013
23.

Boschi D., Sorba G., Bertinaria M., Fruttero R., Calvino R., Gasco A.
Journal of the Chemical Society Perkin Transactions 1,
2001
24.

Gasco A., Mortarini V., Ruà G., Serafino A.
Journal of Heterocyclic Chemistry,
1973
25.

Godovikova T.I., Rakitin O.A., Golova S.P., Vozchikova S.A., Povorin M.V., Khmel'nitskii L.I.
Chemistry of Heterocyclic Compounds,
1994