Abstract
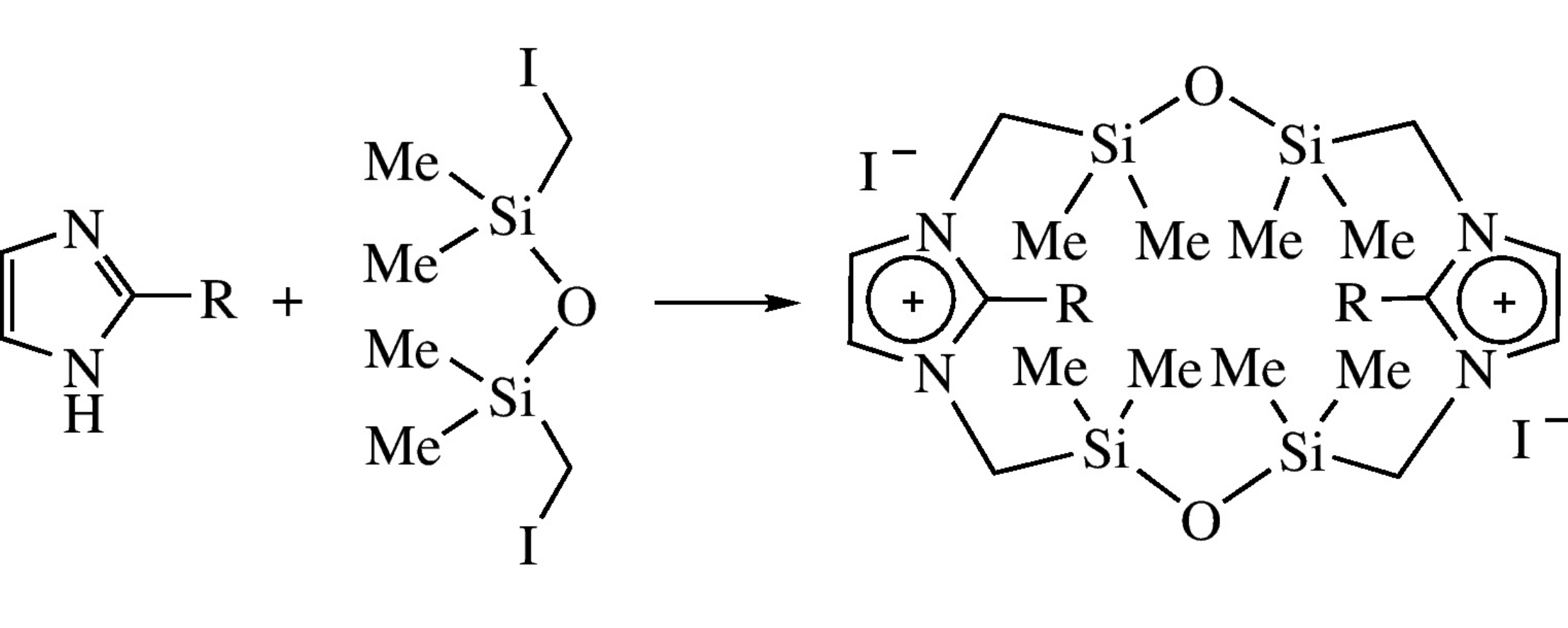
The first representative imidazolophanes bearing dimethylene(tetramethyl)disiloxane spacers were synthesized in one preparative stage by the reaction between imidazoles and 1,3-bis(iodomethyl)-1,1,3,3-tetramethyldisiloxane.
References
1.

Deb M.L., Bhuyan P.J.
Tetrahedron Letters,
2005
2.
![An Efficient Protocol for Henry Reaction Using Basic Ionic Liquid [bmIm]OH as Catalyst and Reaction Medium](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
Wu H., Zhang F., Wan Y., Ye L.
Letters in Organic Chemistry,
2008
3.
![Basic Ionic Liquid as Catalysis and Reaction Medium: A Novel and Green Protocol for the Markovnikov Addition of N-Heterocycles to Vinyl Esters, Using a Task-Specific Ionic Liquid, [bmIm]OH](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Xu J., Liu B., Wu W., Qian C., Wu Q., Lin X.
Journal of Organic Chemistry,
2006
4.

Gong K., Fang D., Wang H., Liu Z.
Monatshefte fur Chemie,
2007
5.

Yadav L.D., Rai A.
Tetrahedron Letters,
2009
6.

Matsuda H., Fukuda N., Ueno T., Katakawa M., Wang X., Watanabe T., Matsui S., Aoyama T., Saito K., Bando T., Matsumoto Y., Nagase H., Matsumoto K., Sugiyama H.
Kidney International,
2011
7.

Shinohara K., Bando T., Sugiyama H.
Anti-Cancer Drugs,
2010
8.

Kanjilal S., Sunitha S., Reddy P.S., Kumar K.P., Murty U.S., Prasad R.B.
European Journal of Lipid Science and Technology,
2009
9.

Chan P.C., Sills R.C., Kissling G.E., Nyska A., Richter W.
Archives of Toxicology,
2007
10.

Hay M.P., Wilson W.R., Denny W.A.
Tetrahedron,
2000
11.

Khabnadideh S., Rezaei Z., Khalafi-Nezhad A., Bahrinajafi R., Mohamadi R., Farrokhroz A.A.
Bioorganic and Medicinal Chemistry Letters,
2003
12.
10.1016/j.mencom.2015.01.012_bib0025
Banerjee
Indian J. Chem., Sect. B,,
2008
13.

Jiang T., Ma X., Zhou Y., Liang S., Zhang J., Han B.
Green Chemistry,
2008
14.
10.1016/j.mencom.2015.01.012_bib0035
Eselev
Klei Germetiki Tekhnologii,
2005
15.

Lee S., Jeon Y., Lim Y., Hossain M.A., Lee S., Cho Y., Ju H., Kim W.
Electrochimica Acta,
2013
16.

Grimison A., Ridd J.H., Smith B.V.
Journal of the Chemical Society (Resumed),
1960
17.

Calvino Casilda V., Pérez-Mayoral E., Bañares M.A., Lozano Diz E.
Chemical Engineering Journal,
2010
18.

Cernatescu C., Comanita E.
Chemical Industry and Chemical Engineering Quarterly,
2009
19.

Díez-Barra E., de la Hoz A., Sánchez-Migallón A., Tejeda J.
Synthetic Communications,
1993
20.

Yarosh N.O., Zhilitskaya L.V., Shagun L.G., Dorofeev I.A., Larina L.I., Voronkov M.G.
Russian Journal of Organic Chemistry,
2013
21.
10.1016/j.mencom.2015.01.012_sbref0055b
Shagun
Russ. J. Org. Chem.,
2013
22.

Reiller P., Mercier-Bion F., Gimenez N., Barré N., Miserque F.
Radiochimica Acta,
2006
23.

Rebek J.
Chemical Society Reviews,
1996
24.
10.1016/j.mencom.2015.01.012_sbref0065b
Gilman
Chem. Rev.,
1997
25.
10.1016/j.mencom.2015.01.012_sbref0065c
Alcalde
Chem. Commun.,
1999
26.

Yoon J., Kim S.K., Singh N.J., Kim K.S.
Chemical Society Reviews,
2006
27.
Kulikov D.V., Karasik A.A., Balueva A.S., Kataeva O.N., Litvinov I.A., Hey-Hawkins E., Sinyashin O.G.
Mendeleev Communications,
2007
28.
10.1016/j.mencom.2015.01.012_sbref0075a
Headley
Aldrichim. Acta,
2007
29.

Ishida Y., Sasaki D., Miyauchi H., Saigo K.
Tetrahedron Letters,
2006
30.

Zvereva O.V., Milyutin A.V., Bobrovskaya O.V., Odegova T.F.
Pharmaceutical Chemistry Journal,
2004
31.

Yu. Ishmuratov G., P. Yakovleva M., R. Mingaleeva G., G. Tolstikov A.
Macroheterocycles,
2011