Abstract
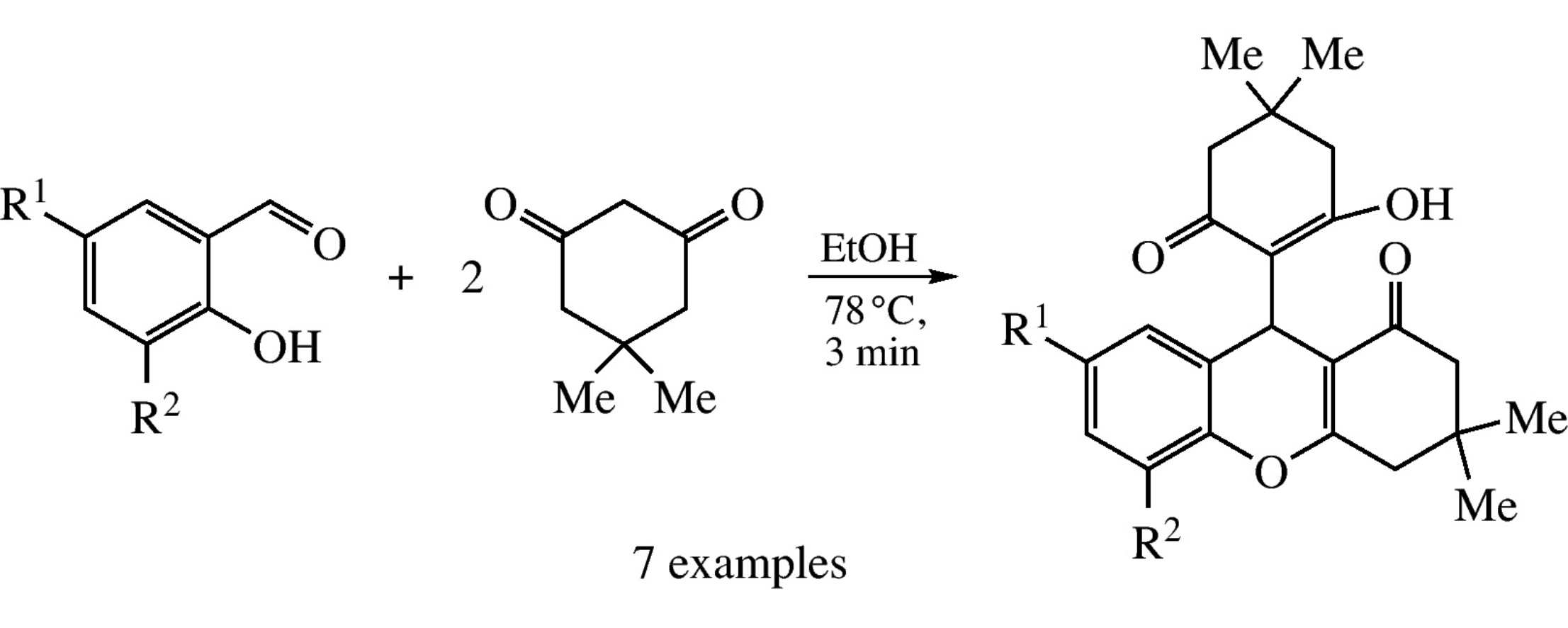
Thermally induced non-catalytic assembling of salicylaldehydes and dimedone in water or ethanol affords 9-(2-hydroxy-4,4-dimethyl-6-oxo-1-cyclohexen-1-yl)-3,3-dimethyl-2,3,4,9-tetrahydro-1H-xanthen-1-ones in 85–95% yields as a sequence of Knoevenagel and Michael reactions.
References
1.

Wahlestedt C., Reis D.J.
Annual Review of Pharmacology and Toxicology,
1993
2.

Blomqvist A.G., Herzog H.
Trends in Neurosciences,
1997
3.

Gerald C., Walker M.W., Criscione L., Gustafson E.L., Batzl-Hartmann C., Smith K.E., Vaysse P., Durkin M.M., Laz T.M., Linemeyer D.L., Schaffhauser A.O., Whitebread S., Hofbauer K.G., Taber R.I., Branchek T.A., et. al.
Nature,
1996
4.

Kanatani A., Ishihara A., Iwaasa H., Nakamura K., Okamoto O., Hidaka M., Ito J., Fukuroda T., MacNeil D.J., Van der Ploeg L.H., Ishii Y., Okabe T., Fukami T., Ihara M.
Biochemical and Biophysical Research Communications,
2000
5.

Mashiko S., Ishihara A., Iwaasa H., Sano H., Oda Z., Ito J., Yumoto M., Okawa M., Suzuki J., Fukuroda T., Jitsuoka M., Morin N.R., MacNeil D.J., Van der Ploeg L.H., Ihara M., et. al.
Endocrinology,
2003
6.

Ishihara A., Kanatani A., Mashiko S., Tanaka T., Hidaka M., Gomori A., Iwaasa H., Murai N., Egashira S., Murai T., Mitobe Y., Matsushita H., Okamoto O., Sato N., Jitsuoka M., et. al.
Proceedings of the National Academy of Sciences of the United States of America,
2006
7.

Thoison O., Hnawia E., Guiéritte-Voegelein F., Sévenet T.
Phytochemistry,
1992
8.

Wang H., Morris-Natschke S.L., Lee K.
Medicinal Research Reviews,
1997
9.

Sabitha G., Arundhathi K., Sudhakar K., Sastry B.S., Yadav J.S.
Synthetic Communications,
2008
10.
10.1016/j.mencom.2015.01.006_bib0050
Li
Chin. J. Org. Chem.,
2005
11.

Nagarapu L., Karnakanti S., Bantu R., Sridhar B.
Synthetic Communications,
2011
12.

Wang X., Shi D., Li Y., Chen H., Wei X., Zong Z.
Synthetic Communications,
2005
13.
![Zn[(L)Proline]2: an eligible candidate for the synthesis of xanthenediones in water](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kidwai M., Jain A.
Applied Organometallic Chemistry,
2012
14.

Zhang P., Yu Y., Zhang Z.
Synthetic Communications,
2008
15.

Sheldon R.A.
Journal of Molecular Catalysis A Chemical,
1996
16.

Pore D.M., Shaikh T.S., Undale K.A., Gaikwad D.S.
Comptes Rendus Chimie,
2010
17.
Elinson M.N., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2012
18.

19.
![Stereoselective electrocatalytic transformations of malononitrile and aromatic aldehydes into (1R,5S,6R)*-4,4-dialkoxy-2-amino-6-aryl-1,5-dicyano-3-azabicyclo[3.1.0]hex-2-enes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Feducovich K., Zaimovskaya T.A., Vereshchagin A.N., Nikishin G.I.
Russian Chemical Bulletin,
2005
20.

Elinson M.N., Vereshchagin A.N., Feducovich S.K., Zaimovskaya T.A., Starikova Z.A., Belyakov P.A., Nikishin G.I.
Tetrahedron Letters,
2007
21.

Liu W., Jiang H., Zhu S., Wang W.
Tetrahedron,
2009
22.
Demchuk D.V., Elinson M.N., Nikishin G.I.
Mendeleev Communications,
2011
23.

Patai S., Israeli Y.
Journal of the Chemical Society (Resumed),
1960