Abstract
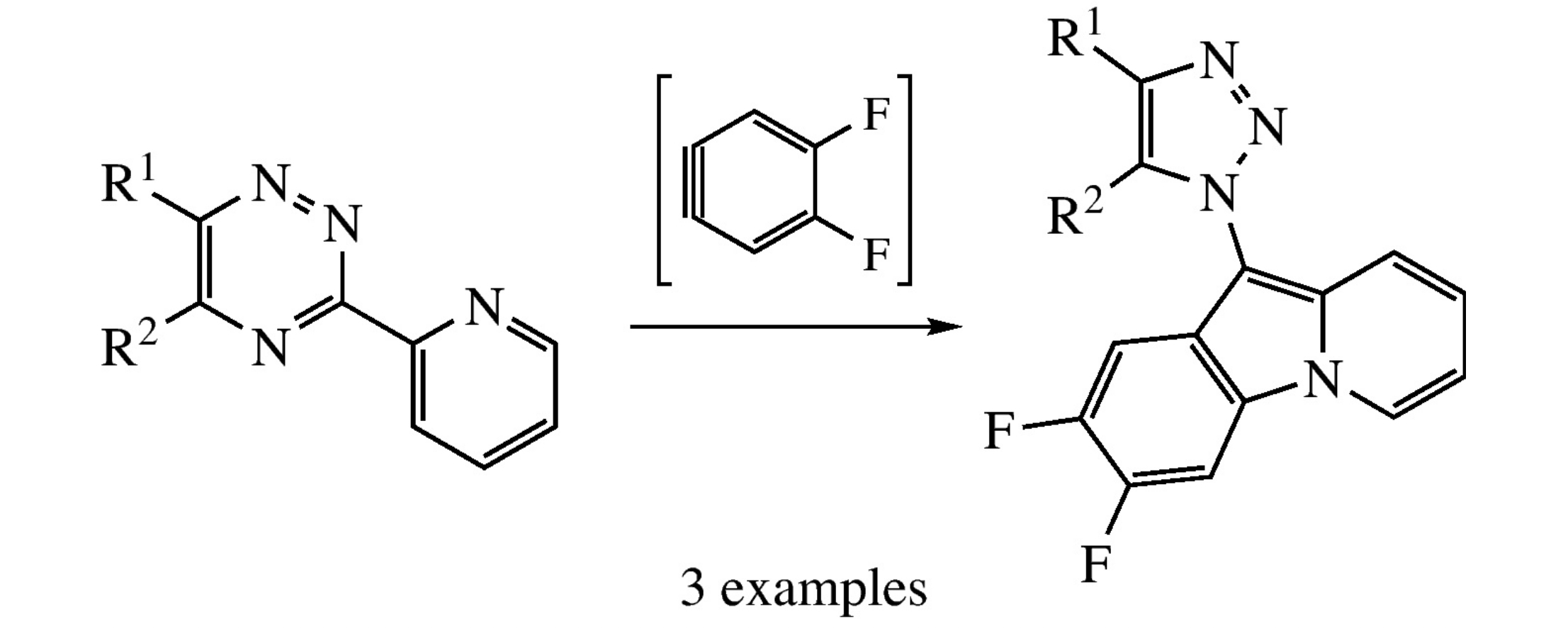
Reaction between 3-(2-pyridyl)-1,2,4-triazines and in situ generated 4,5-difluorobenzyne in toluene affords 2,3-difluoro-10-(1H-1,2,3-triazol-1-yl)pyrido[1,2-a]indoles. The structure of one representative compound was confirmed by X-ray diffraction analysis.
References
1.
![Synthesis and Antitumor Activity of Methoxy-indolo[2,1-a]isoquinolines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ambros R., Angerer S.V., Wiegrebe W.
Archiv der Pharmazie,
1988
2.
![Indolo[2,1-a]isoquinolines. Syntheses, steroid hormone receptor binding affinities, and cytostatic activity](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ambros R., Schneider M.R., Von Angerer S.
Journal of Medicinal Chemistry,
1990
3.
T. W. Hudyma, X. Zheng, F. He, M. Ding, C.P. Bergstrom, P. Hewawasam, S. W. Martin and R. G. Gentles, U.S. Pat. Appl. Publ., 2006, 318, Cont.- In-Part of U.S. Ser. No. 181, 639.(Chem. Abstr., 2006, 145, 189063).
4.

Orlemans E.O., Verboom W., Scheltinga M.W., Reinhoudt D.N., Lelieveld P., Fiebig H.H., Winterhalter B.R., Double J.A., Bibby M.C.
Journal of Medicinal Chemistry,
1989
5.

Dong S., Wang W., Yin S., Li C., Lu J.
Synthetic Metals,
2009
6.

Trashakhova T.V., Nosova E.V., Valova M.S., Slepukhin P.A., Lipunova G.N., Charushin V.N.
Russian Journal of Organic Chemistry,
2011
7.

Lipunova G.N., Nosova E.V., Laeva A.A., Charushin V.N.
Pharmaceutical Chemistry Journal,
2012
8.

Kang I., Shim H., Zyung T.
Chemistry of Materials,
1997
9.

Gurge R.M., Sarker A.M., Lahti P.M., Hu B., Karasz F.E.
Macromolecules,
1997
10.
![Synthesis of Pyrido[1,2-a]indole Malonates and Amines through Aryne Annulation](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rogness D.C., Markina N.A., Waldo J.P., Larock R.C.
Journal of Organic Chemistry,
2012
11.
![Palladium and Copper Cocatalyzed Tandem N–H/C–H Bond Functionalization: Synthesis of CF3-Containing Indolo- and Pyrrolo[2,1-a]isoquinolines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Sun L., Liao Z., Tang R., Deng C., Zhang X.
Journal of Organic Chemistry,
2012
12.
Sukhanov G.T., Sakovich G.V., Filippova Y.V., Bagryanskaya I.Y., Sukhanova A.G.
Mendeleev Communications,
2014
13.

Agalave S.G., Maujan S.R., Pore V.S.
Chemistry - An Asian Journal,
2011
14.

Kim H., Lee S., Lee J., Tae J.
Organic Letters,
2010
15.

Ghosh K., Kar D., Joardar S., Sahu D., Ganguly B.
RSC Advances,
2013
16.

Bhalla V., Singh H., Kumar M.
Dalton Transactions,
2012
17.

Braun J.V., Nelles J.
Berichte der deutschen chemischen Gesellschaft (A and B Series),
1937
18.

Deegan A., Rose F.L.
Journal of the Chemical Society C Organic,
1971
19.

Naredla R.R., Zheng C., Nilsson Lill S.O., Klumpp D.A.
Journal of the American Chemical Society,
2011
20.
10.1016/j.mencom.2015.01.003_sbref0065d
Soldatenkov
J. Org. Chem. USSR,
1982
21.
![FLASH VACUUM PYROLYSIS OF 2-BENZYLPYRIDINEN-OXIDES. SYNTHESIS OF METHYLPYRIDO[1,2-a]INDOLES](/storage/images/resized/yNSijlgQghQF53VZuyFLA30CKDe4j3HK74Vtpnxa_small_thumb.webp)
Ohsawa A., Kawaguchi T., Igeta H.
Chemistry Letters,
1981
22.
L. M. Gaster and P. A. Wyman, Patent US 5998409A, 1995 (Chem. Abstr., 1995, 122, 265390).
23.
![Benzyne-mediated rearrangement of 3-(2-pyridyl)-1,2,4-triazines into 10-(1H-1,2,3-triazol-1-yl)pyrido[1,2-a]indoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Nikonov I.L., Kopchuk D.S., Kovalev I.S., Zyryanov G.V., Khasanov A.F., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2013
24.

Kovalev I.S., Kopchuk D.S., Zyryanov G.V., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2012
25.

Dubrovskiy A.V., Markina N.A., Larock R.C.
Organic and Biomolecular Chemistry,
2013
26.

Gebara K.S., Casagrande G.A., Raminelli C.
Tetrahedron Letters,
2011
27.
10.1016/j.mencom.2015.01.003_sbref0080b
Castedo
Angew. Chem., Int. Ed. Engl.,
1997
28.

Raminelli C., Liu Z., Larock R.C.
Journal of Organic Chemistry,
2006
29.

Luo R., Liao J., Xie L., Tang W., Chan A.S.
Chemical Communications,
2013
30.

Bula R.P., Oppel I.M., Bettinger H.F.
Journal of Organic Chemistry,
2012
31.

Webster R., Böing C., Lautens M.
Journal of the American Chemical Society,
2008
32.

Layeva A.A., Nosova E.V., Lipunova G.N., Charushin V.N.
Russian Chemical Bulletin,
2008
33.
![The reaction of acetic acid 2-selenoxo-2H-pyridin-1-yl esters with benzynes: A convenient route to Benzo[b]seleno[2,3-b]pyridines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Narasimha Rao U., Sathunuru R., Maguire J.A., Biehl E.
Journal of Heterocyclic Chemistry,
2004
34.

Hodgson D.M., Bebbington M.W., Willis P.
Organic and Biomolecular Chemistry,
2003
35.

Hodgson D.M., Bebbington M.W., Willis P.
Organic Letters,
2002
36.
F. R. Busch, S.J. Hecker, P.R. McGuirk, B.T. O’Neill and H. A. Watson, Jr., Patent EP0342849, 1989 (Chem. Abstr., 1990, 113, 406177).
37.

Case F.H.
Journal of Organic Chemistry,
1965
38.

Kozhevnikov V.N., Shabunina O.V., Kopchuk D.S., Ustinova M.M., König B., Kozhevnikov D.N.
Tetrahedron,
2008
39.

Kopchuk D.S., Zyryanov G.V., Kovalev I.S., Khasanov A.F., Medvedevskikh A.S., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2013
40.

Dalvit C., Vulpetti A.
ChemMedChem,
2012
41.

Dunitz J.D., Taylor R.
Chemistry - A European Journal,
1997