Abstract
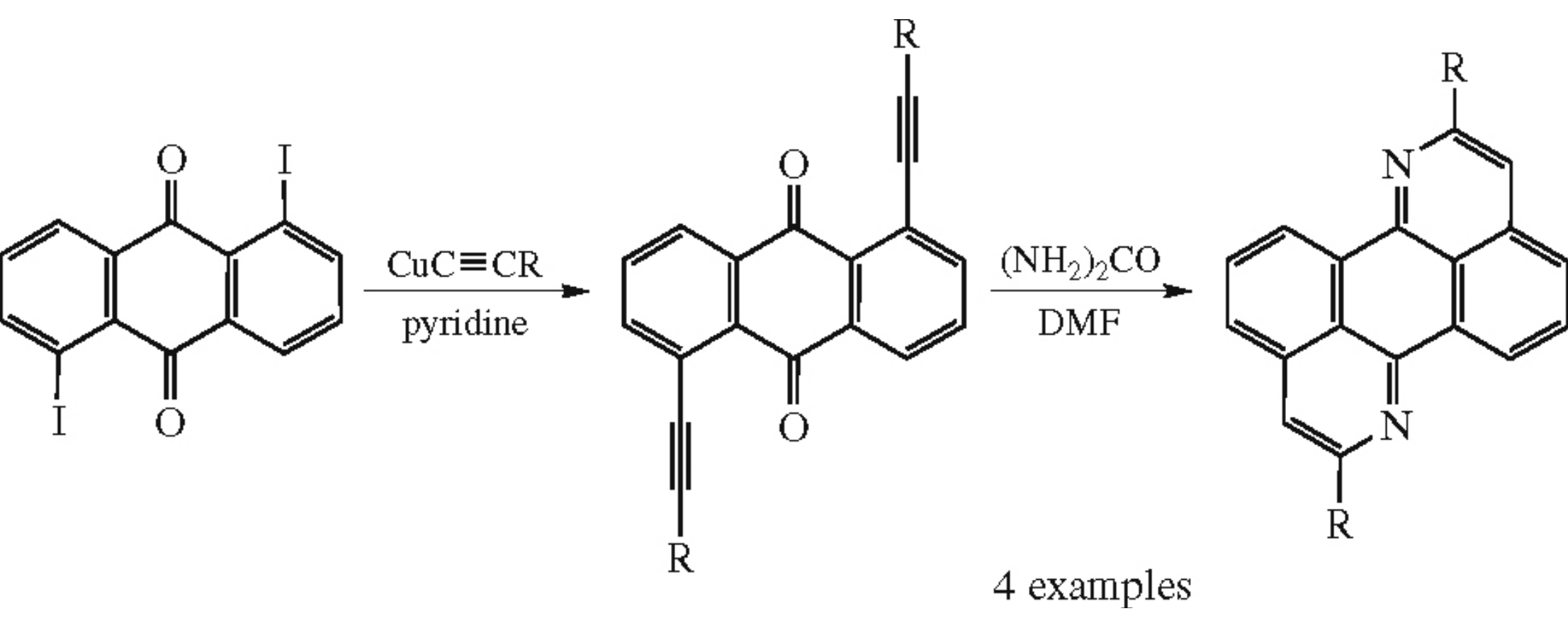
Benzo[de]isoquino[1,8-gh]quinolines were prepared by the reaction between 1,5-diethynyl-9,10-anthraquinones and urea in DMF. Light-induced electron transfer from regioregular poly(3-hexylthiophene) to the synthesized molecules was detected by an EPR spectroscopy technique.
References
1.
Organic Photovoltaics: Concepts and Realization, eds. C. J. Brabec, V. Dyakonov, J. Parisi and N. S. Sariciftci, Springer, Berlin, 2010.
2.

Chochos C.L., Tagmatarchis N., Gregoriou V.G.
RSC Advances,
2013
3.

Kozma E., Catellani M.
Dyes and Pigments,
2013
4.
Mumyatov A.V., Leshanskaya L.I., Anokhin D.V., Dremova N.N., Troshin P.A.
Mendeleev Communications,
2014
5.
![689. Syntheses of polycyclic systems. Part II. 8-Ketodibenzo[c,mn]-acridine and 8 : 16-diazadibenzo[b,k]perylene](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Braude E.A., Fawcett J.S.
Journal of the Chemical Society (Resumed),
1951
6.

Galt R.H., Loudon J.D., Sloan A.D.
Journal of the Chemical Society (Resumed),
1958
7.

Naumann C., Langhals H.
Chemische Berichte,
1990
8.
H. Langhals and C. Naumann, German Patent 4005056, 1991.
9.
F. Bienewald, B. Schmidhalter, U. Berens, H.J. Kirner, US Patent WO 2006/120143, 2006.
10.
F. Bienewald, B. Schmidhalter, U. Berens and H. J. Kirner, US Patent US 2009/101890, 2009.
11.
![Urea as an organic solvent and reagent for the addition/cyclization/fragmentation cascades leading to 2-R-7H-dibenzo[de,h]quinolin-7-one analogues of Aporphinoid alkaloids](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Baranov D.S., Vasilevsky S.F., Gold B., Alabugin I.V.
RSC Advances,
2011
12.

Moroz A.A., Piskunov A.V., Shvartsberg M.S.
Russian Chemical Bulletin,
1981
13.

Murata M., Yamada M., Fujita T., Kojima K., Kurihara M., Kubo K., Kobayashi Y., Nishihara H.
Journal of the American Chemical Society,
2001
14.

Gouloumis A., González-Rodríguez D., Vázquez P., Torres T., Liu S., Echegoyen L., Ramey J., Hug G.L., Guldi D.M.
Journal of the American Chemical Society,
2006
15.

Li F., Cheng J., Chai X., Jin S., Wu X., Yu G., Liu S.H., Chen G.Z.
Organometallics,
2011
16.

Seidel N., Hahn T., Liebing S., Seichter W., Kortus J., Weber E.
New Journal of Chemistry,
2013
17.
10.1016/j.mencom.2014.11.026_bib0040
Castro
J. Org. Chem.,
1963
18.

Scholl R., Hass S., Meyer H.K.
Berichte der deutschen chemischen Gesellschaft (A and B Series),
1929
19.

Marrocchi A., Lanari D., Facchetti A., Vaccaro L.
Energy and Environmental Science,
2012
20.

Chen T., Wu X., Rieke R.D.
Journal of the American Chemical Society,
1995
21.

Sariciftci N.S., Smilowitz L., Heeger A.J., Wudl F.
Science,
1992
22.

Marumoto K., Fujimori T., Ito M., Mori T.
Advanced Energy Materials,
2012
23.

Marumoto K., Muramatsu Y., Nagano Y., Iwata T., Ukai S., Ito H., Kuroda S., Shimoi Y., Abe S.
Journal of the Physical Society of Japan,
2005