Abstract
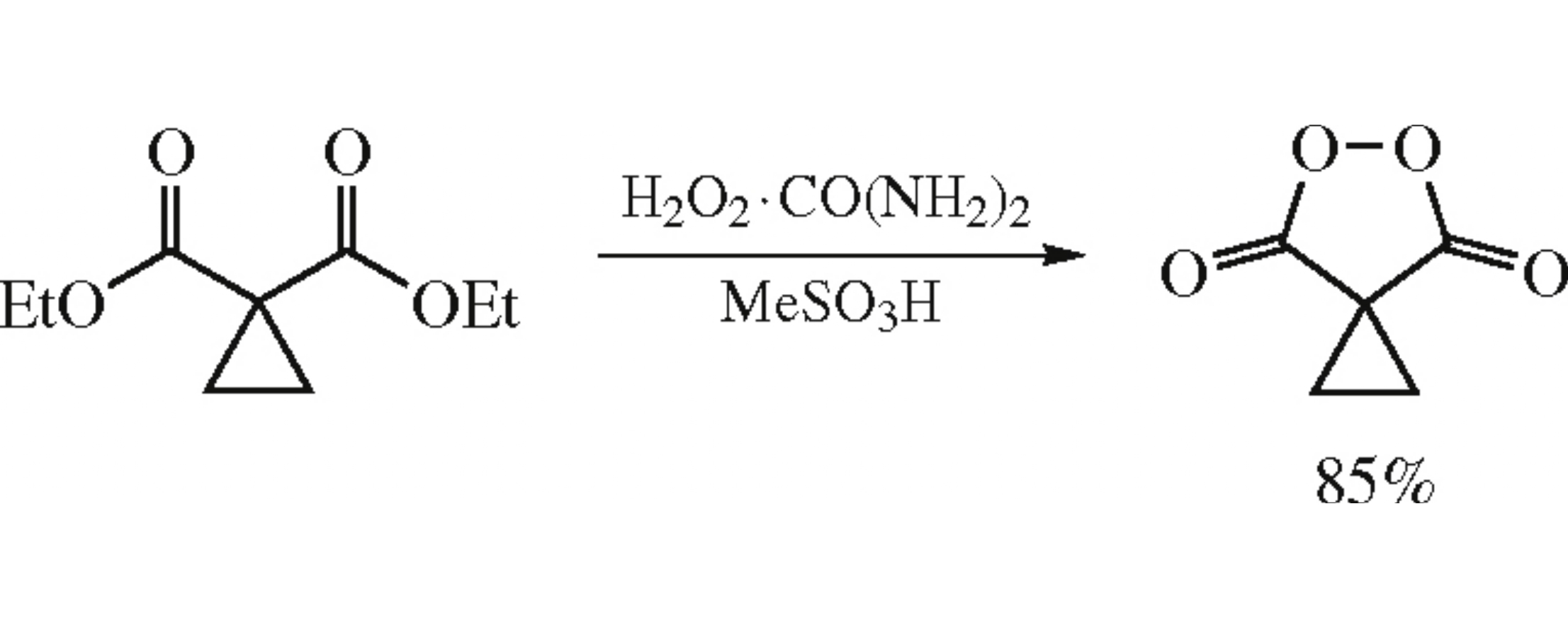
Cyclopropane-1,1-dicarbonyl peroxide was prepared in 85% yield by the reaction of diethyl cyclopropane-1,1-dicarboxylate with the urea hydrogen peroxide clathrate in the presence of methanesulfonic acid.
References
1.

Adam W., Diehl J.W.
Journal of the Chemical Society Chemical Communications,
1972
2.

Perrin C.L., Arrhenius T.
Journal of the American Chemical Society,
1978
3.

Greene F.D.
Journal of the American Chemical Society,
1956
4.

Greene F.D.
Journal of the American Chemical Society,
1956
5.

Greene F.D.
Journal of the American Chemical Society,
1959
6.

Greene F.D., Rees W.W.
Journal of the American Chemical Society,
1960
7.

Greene F.D., Rees W.W.
Journal of the American Chemical Society,
1960
8.

Griffith J.C., Jones K.M., Picon S., Rawling M.J., Kariuki B.M., Campbell M., Tomkinson N.C.
Journal of the American Chemical Society,
2010
9.

Schwarz M., Reiser O.
Angewandte Chemie - International Edition,
2011
10.

Yuan C., Axelrod A., Varela M., Danysh L., Siegel D.
Tetrahedron Letters,
2011
11.

Picon S., Rawling M., Campbell M., Tomkinson N.C.
Organic Letters,
2012
12.

Jones K.M., Tomkinson N.C.
Journal of Organic Chemistry,
2012
13.

Rawling M.J., Rowley J.H., Campbell M., Kennedy A.R., Parkinson J.A., Tomkinson N.C.
Chemical Science,
2014
14.

Rawling M.J., Tomkinson N.C.
Organic and Biomolecular Chemistry,
2013
15.

Yuan C., Liang Y., Hernandez T., Berriochoa A., Houk K.N., Siegel D.
Nature,
2013
16.
R. K. Singh and S. Danishefsky, J. Org. Chem., 1975, 40, 2969.
17.
R. K. Singh and S. Danishefsky, Org. Synth. Coll. Vol., 1981, 7, 411.
18.

Heiszman J., Bitter I., Harsányi K., Töke L.
Synthesis,
1987
19.
K. M. Jones, PhD Thesis, Cardiff University, 2010.
20.

Dmowski W., Wolniewicz A.
Journal of Fluorine Chemistry,
2000
21.
W. Adam and L. Szendrey, Tetrahedron Lett., 1972, 26, 2669.
22.
C. Singh, N.C. Srivastav, N. Srivastava and S. K. Puri, Tetrahedron Lett., 2005, 46, 2757.
23.
E. H. White, D.F. Roswell, A.C. Dupont and A. A. Wilson, J. Am. Chem. Soc., 1987, 109, 5189.
24.
P. Dussault and I. Q. Lee, J. Org. Chem., 1992, 57, 1952.
25.
D. L. Yin, P. Bernhardt, K.L. Morley, Y. Jiang, J.D. Cheeseman, V. Purpero, J.D. Schrag and R. J. Kazlauskas, Biochemistry, 2010, 49, 1931.
26.
L. Wiermans, S. Hofzumahaus, C. Schotten, L. Weigand, M. Schallmey, A. Schallmey and P. Domínguez de María, ChemCatChem, 2013, 5, 3719.
27.

Eliasen A.M., Thedford R.P., Claussen K.R., Yuan C., Siegel D.
Organic Letters,
2014
28.
10.1016/j.mencom.2014.11.010_sbref0040b
Rappoport
The Chemistry of Peroxides,
2007