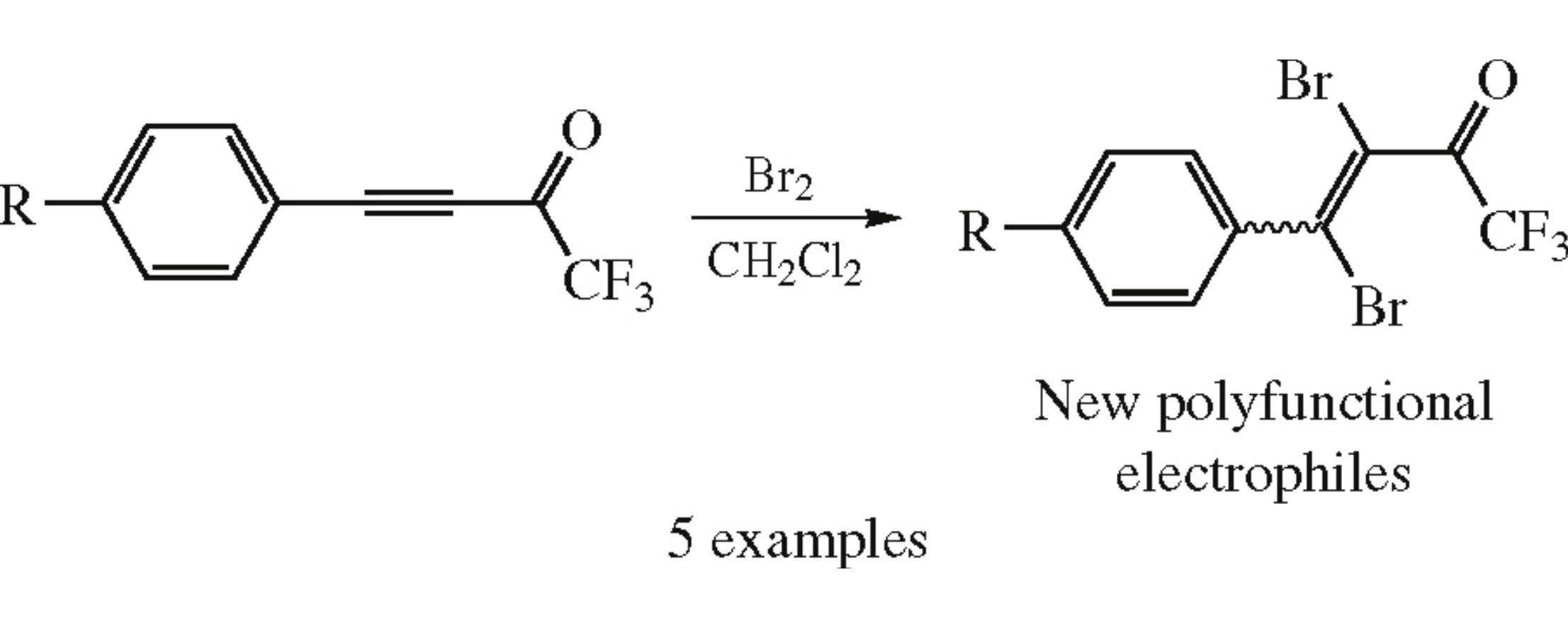
Abstract
Bromination of 4-aryl-1,1,1-trifluorobut-3-yn-2-ones gives 4-aryl-3,4-dibromo-1,1,1-trifluorobut-3-en-2-ones whose reactivity towards N-nucleophiles (hydrazine and ethylenediamine) was investigated.
References
1.
10.1016/j.mencom.2014.11.009_sbref0005a
Kirsch
Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications,
2004
3.
10.1016/j.mencom.2014.11.009_sbref0005c
Bégué
Bioorganic and Medicinal Chemistry of Fluorine,
2008
4.
Fluorine and Health. Molecular Imaging, Biomedical Materials and Pharmaceuticals, eds. A. Tressaud and G. Haufe, Elsevier, Amsterdam, 2008, pp. 553-778.
5.

Nenajdenko V., Haufe G., Muzalevskiy V., Shastin A., Balenkova E.
Synthesis,
2009
7.

Nenajdenko V., Serdyuk O., Abaev V., Butin A.
Synthesis,
2011
8.
O. V. Serdyuk, V.M. Muzalevskiy and V. G. Nenajdenko, Synthesis, 2012, 2115.
9.
T. Liang, C.N. Neumann and T. Ritter, Angew. Chem. Int. Ed., 2013, 52, 8214.
10.
J. Wang, M. Sanchez-Rosello, J.L. Acena, C. del Pozo, A.E. Sorochinsky, S. Fustero, V.A. Soloshonok and H. Liu, Chem. Rev., 2014, 114, 2432.
11.
I. Ojima, J. Org. Chem., 2013, 78, 6358.
12.
Fluorine in Heterocyclic Chemistry, ed. V. G. Nenajdenko, Springer, 2014.
13.
![Synthesis and Biological Evaluation of the 1,5-Diarylpyrazole Class of Cyclooxygenase-2 Inhibitors: Identification of 4-[5-(4-Methylphenyl)-3- (trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide (SC-58635, Celecoxib)](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Penning T.D., Talley J.J., Bertenshaw S.R., Carter J.S., Collins P.W., Docter S., Graneto M.J., Lee L.F., Malecha J.W., Miyashiro J.M., Rogers R.S., Rogier D.J., Yu S.S., Anderson G.D., Burton E.G., et. al.
Journal of Medicinal Chemistry,
1997
14.

Habeeb A.G., Praveen Rao P.N., Knaus E.E.
Journal of Medicinal Chemistry,
2001
15.

Singh S.K., Vobbalareddy S., Shivaramakrishna S., Krishnamraju A., Rajjak S.A., Casturi S.R., Akhila V., Rao Y.K.
Bioorganic and Medicinal Chemistry Letters,
2004
16.

Singh S.K., Reddy P.G., Rao K.S., Lohray B.B., Misra P., Rajjak S.A., Rao Y.K., Venkateswarlu A.
Bioorganic and Medicinal Chemistry Letters,
2004
17.
![2-Hydroxymethyl-4-[5-(4-methoxyphenyl)-3-trifluoromethyl-1H-1-pyrazolyl]-1-benzenesulfonamide (DRF-4367): an orally active COX-2 inhibitor identified through pharmacophoric modulation](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Singh S.K., Vobbalareddy S., Kalleda S.R., Rajjak S.A., Casturi S.R., Datla S.R., Mamidi R.N., Mullangi R., Bhamidipati R., Ramanujam R., Akella V., Yeleswarapu K.R.
Organic and Biomolecular Chemistry,
2004
18.

Chowdhury M.A., Abdellatif K.R., Dong Y., Das D., Suresh M.R., Knaus E.E.
Journal of Medicinal Chemistry,
2009
19.
V. G. Nenaidenko and E. S. Balenkova, Zh. Org. Khim., 1992, 28, 600 (in Russian).
20.

Nenajdenko V.G., Leshcheva I.F., Balenkova E.S.
Tetrahedron,
1994
21.

Nenajdenko V., Krasovsky A., Lebedev M., Balenkova E.
Synlett,
2000
22.

Krasovsky A.L., Nenajdenko V.G., Balenkova E.S.
Tetrahedron,
2001
23.

Nenajdenko V.G., Krasovskiy A.L., Balenkova E.S.
Tetrahedron,
2007
24.
A. Yu. Rulev, V.M. Muzalevskiy, E.V. Kondrashov, I.A. Ushakov, A.R. Romanov, V.N. Khrustalev and V. G. Nenajdenko, Org. Lett., 2013, 15, 2726.
25.

Nenajdenko V., Sanin A., Balenkova E.
Molecules,
1997
26.

Nenajdenko V.G., Sanin A.V., Balenkova E.S.
Russian Chemical Reviews,
1999
27.
S. V. Druzhinin, E.S. Balenkova and V. G. Nenajdenko, Tetrahedron, 2007, 63, 7753.
28.
Nenajdenko V.G., Balenkova E.S.
Arkivoc,
2011
29.

Fustero S., Sánchez-Roselló M., Barrio P., Simón-Fuentes A.
Chemical Reviews,
2011
30.

Fustero S., Simón-Fuentes A., Sanz-Cervera J.F.
Organic Preparations and Procedures International,
2009
31.

Sasaki S., Ikekame Y., Tanayama M., Yamauchi T., Higashiyama K.
Synlett,
2012
32.

Linderman R.J., Kirollos K.S.
Tetrahedron Letters,
1989
33.

Jones B.G., Branch S.K., Thompson A.S., Threadgill M.D.
Journal of the Chemical Society Perkin Transactions 1,
1996
34.

Nenajdenko V.G., Muzalevskiy V.M., Shastin A.V., Balenkova E.S., Kondrashov E.V., Ushakov I.A., Rulev A.Y.
Journal of Organic Chemistry,
2010
35.

Roy S., Davydova M.P., Pal R., Gilmore K., Tolstikov G.A., Vasilevsky S.F., Alabugin I.V.
Journal of Organic Chemistry,
2011
36.

Jeon S.L., Choi J.H., Kim B.T., Jeong I.H.
Journal of Fluorine Chemistry,
2007
37.

Martins M.A., Beck P.H., Moreira D.N., Buriol L., Frizzo C.P., Zanatta N., Bonacorso H.G.
Journal of Heterocyclic Chemistry,
2010
38.
10.1016/j.mencom.2014.11.009_bib0065
Schuh
Synthesis,
2007