Abstract
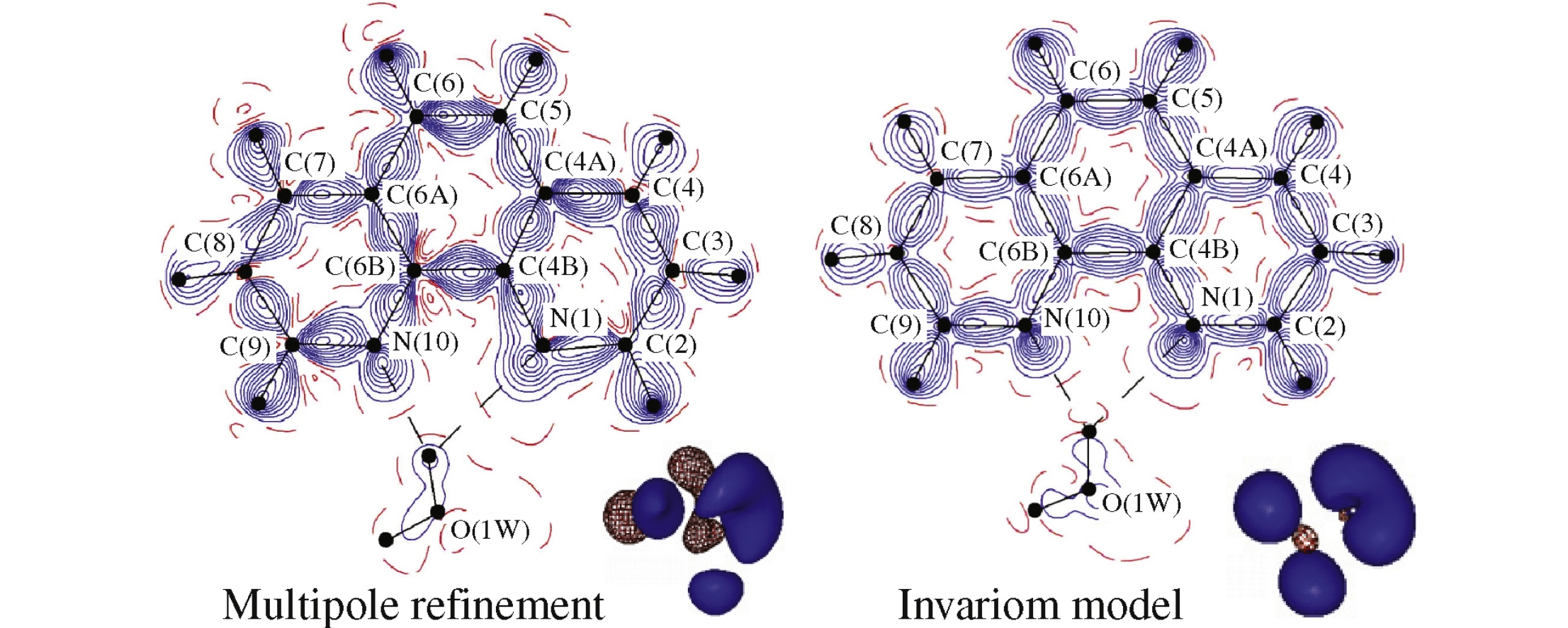
The modeling of experimental electron density in a twinned crystal of 1,10-phenanthroline hydrate within an invariom approach revealed its another advantage for charge density studies, which is assessing the reliability of chemically relevant information provided by a conventional multipole refinement against high-resolution X-ray diffraction data.
References
1.
10.1016/j.mencom.2014.09.013_bib0005
Gatti
Modern Charge-Density Analysis,
2012
2.

Dittrich B., Koritsánszky T., Luger P.
Angewandte Chemie - International Edition,
2004
3.
10.1016/j.mencom.2014.09.013_bib0015
Dittrich
2013
4.

Holstein J.J., Hübschle C.B., Dittrich B.
CrystEngComm,
2012
5.

Dittrich B., Hübschle C.B., Holstein J.J., Fabbiani F.P.
Journal of Applied Crystallography,
2009
6.
10.1016/j.mencom.2014.09.013_bib0030
Schurmann
2012
7.

Luger P., Weber M., Hübschle C., Tacke R.
Organic and Biomolecular Chemistry,
2013
8.
10.1016/j.mencom.2014.09.013_bib0040
Dittrich
2009
9.

Dittrich B., Koritsanszky T., Volkov A., Mebs S., Luger P.
Angewandte Chemie - International Edition,
2007
10.
10.1016/j.mencom.2014.09.013_bib0050
Holstein
2010
11.
10.1016/j.mencom.2014.09.013_bib0055
Tian
1995
12.

Sammes P.G., Yahioglu G.
Chemical Society Reviews,
1994
13.

Binnemans K., Lenaerts P., Driesen K., Görller-Walrand C.
Journal of Materials Chemistry A,
2004
14.

Li L., Lin K., Ho C., Sun C., Yang H.
Chemical Communications,
2006
15.

Accorsi G., Listorti A., Yoosaf K., Armaroli N.
Chemical Society Reviews,
2009
16.

Dittrich B., Spackman M.A.
Acta Crystallographica Section A Foundations of Crystallography,
2007
17.
10.1016/j.mencom.2014.09.013_bib0085
Kalinowski
2007
18.

Weber M., Grabowsky S., Hazra A., Naskar S., Banerjee S., Mondal N.B., Luger P.
Chemistry - An Asian Journal,
2011
19.
10.1016/j.mencom.2014.09.013_bib0095
Bacsa
2013
20.
10.1016/j.mencom.2014.09.013_bib0100
Dadda
2012
21.
10.1016/j.mencom.2014.09.013_bib0105
Bader
Atoms in Molecules. A Quantum Theory,
1990
22.

Espinosa E., Molins E., Lecomte C.
Chemical Physics Letters,
1998
23.

Espinosa E., Alkorta I., Rozas I., Elguero J., Molins E.
Chemical Physics Letters,
2001
24.
Lyssenko K.A., Korlyukov A.A., Antipin M.Y.
Mendeleev Communications,
2005
25.

Borissova A.O., Korlyukov A.A., Antipin M.Y., Lyssenko K.A.
Journal of Physical Chemistry A,
2008
26.

Puntus L.N., Lyssenko K.A., Antipin M.Y., Bünzli J.G.
Inorganic Chemistry,
2008
27.
Lyssenko K.A.
Mendeleev Communications,
2012
28.

Chęcińska L., Mebs S., Hübschle C.B., Förster D., Morgenroth W., Luger P.
Organic and Biomolecular Chemistry,
2006
29.

Lyssenko K.A., Nelyubina Y.V., Kostyanovsky R.G., Antipin M.Y.
ChemPhysChem,
2006
30.

Nelyubina Y.V., Antipin M.Y., Cherepanov I.A., Lyssenko K.A.
CrystEngComm,
2010
31.

Hirshfeld F.L.
Acta Crystallographica Section A,
1976