Abstract
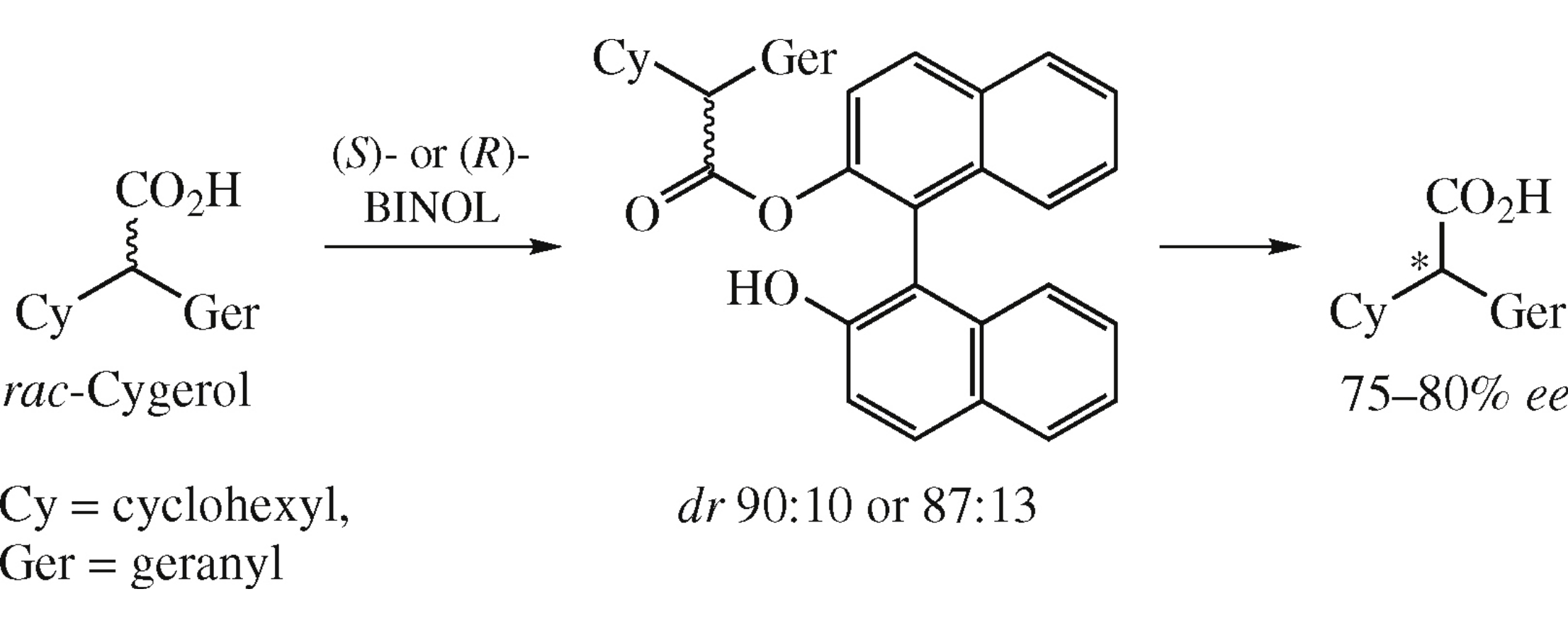
A kinetic resolution of racemic (cyclohexyl)(geranyl)acetic acid, the active ingredient of wound-curing medication Cygerol, to (S)- and (R)-enantiomers was achieved by diastereoselective esterification with (S)- or (R)-BINOL.
References
1.
L. S. Leder-Pakkendorf, Dokl. Akad. Nauk SSSR, 1941, 31, 757 (in Russian).
2.
A. G. Noskov, Meditsinskaya Radiologiya, 1967, 12, 55 (in Russian).
3.
L. S. Bondar and R. A. Okunev, Brit. Pat. GB 1,173,419 (Chem. Abstr., 1970, 72, 54770f).
4.
L. S. Bondar and R. A. Okunev, Ger. Offen. DE 1,801,868 (Chem. Abstr., 1970, 73, 34814r).
5.
L. S. Bondar and R. A. Okunev, Author's Sertificate 333162 USSR (Chem. Abstr., 1972, 77, 100910e).
6.

Cesaro A., Russo E., Crescenzi V.
The Journal of Physical Chemistry,
1976
7.
J. Lapienis and V. J. Zaras, Lith. Pat. LT 4,382 (Chem. Abstr., 1999, 130, 287048h).
8.
B. B. Smirnov, G.V. Kryshtal, A.G. Konopljannikov, S.G. Zlotin and G. M. Zhdankina, RF Patent 2301667 (Chem. Abstr., 2007, 147, 110262).
9.
(a) Chirality in Drug Research, eds. E. Francotte and W. Lindner, Wiley- VCH, Weinheim, 2006; (b) Chirality in Drug Design and Develop- ment, eds. I. K. Reddy and R. Mehvar, Marcel Decker, New York, 2004; (c) H. Caner, E. Groner, L. Levy and I. Agranat, Drug Discov. Today, 2004, 9, 105.
10.
10.1016/j.mencom.2014.09.002_sbref0050a
Rodriguez-Docampo
Chem. Cat. Chem.,
2012
11.

Díaz de Villegas M.D., Gálvez J.A., Etayo P., Badorrey R., López-Ram-de-Víu P.
Chemical Society Reviews,
2011
12.

Shiina I., Nakata K., Ono K., Onda Y., Itagaki M.
Journal of the American Chemical Society,
2010
13.

Mazón A., Nájera C., Yus M., Heumann A.
Tetrahedron Asymmetry,
1992
14.

Tanaka F., Node M., Tanaka K., Mizuchi M., Hosoi S., Nakayama M., Taga T., Fuji K.
Journal of the American Chemical Society,
1995
15.

Hu X., Shan Z., Chang Q.
Tetrahedron Asymmetry,
2012