Abstract
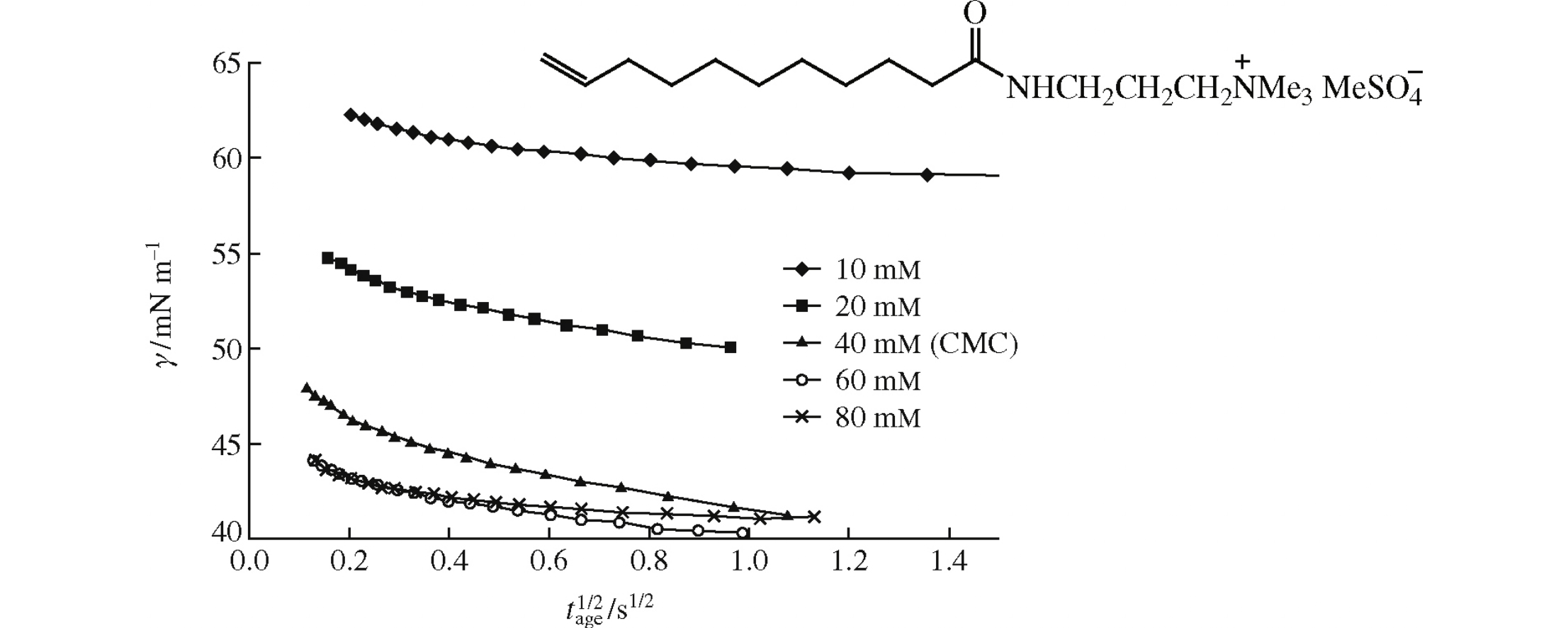
The dynamic surface tension and adsorption properties of the aqueous solutions of N’-undecylenamidopropyl-N”-trimethylammonium methyl sulfate, an eco-friendly cationic surfactant, were evaluated by a maximum bubble pressure method at various life times.
References
1.
10.1016/j.mencom.2014.04.022_bib0005
Pletnev
SOFW Journal,
2006
2.
Surfactants from Renewable Resources, eds. M. Kjellin and I. Johansson, John Wiley & Sons, Chichester, UK, 2010.
3.

Pletnev M.Y.
Characterization of Liquids, Nano- and Microparticulates, and Porous Bodies Using Ultrasound,
2001
4.

Summers M., Eastoe J.
Advances in Colloid and Interface Science,
2003
5.

Van der Steen M., Stevens C.
ChemSusChem,
2009
6.
R ewocid UTM 185 – Mild Quaternary Surfactant, Booklet, Evonik Goldschmidt, Essen, Germany, Feb. 2008.
7.

Fainerman V.B., Mys V.D., Makievski A.V., Petkov J.T., Miller R.
Journal of Colloid and Interface Science,
2006
8.
V. B. Fainerman and R. Miller, in Progress in Colloid and Interface Science, eds. R. Miller and L. Liggieri, Koninklijke Brill NV, Leiden, 2011, vol. 2, pp. 75-118.
9.
K. J. Mysels and A. T. Florence, in Clean Surfaces: Their Preparation and Characterization for Interfacial Studies, ed. G. Goldfinger, Marcel Dekker, New York, 1970, pp. 227-268.
10.

Christov N.C., Danov K.D., Kralchevsky P.A., Ananthapadmanabhan K.P., Lips A.
Langmuir,
2006
11.

Parker R.A., Wasik S.P.
The Journal of Physical Chemistry,
1959
12.

Danov K.D., Kralchevsky P.A., Ananthapadmanabhan K.P., Lips A.
Journal of Colloid and Interface Science,
2006
13.

Ritacco H., Langevin D., Diamant H., Andelman D.
Langmuir,
2011
14.