Abstract
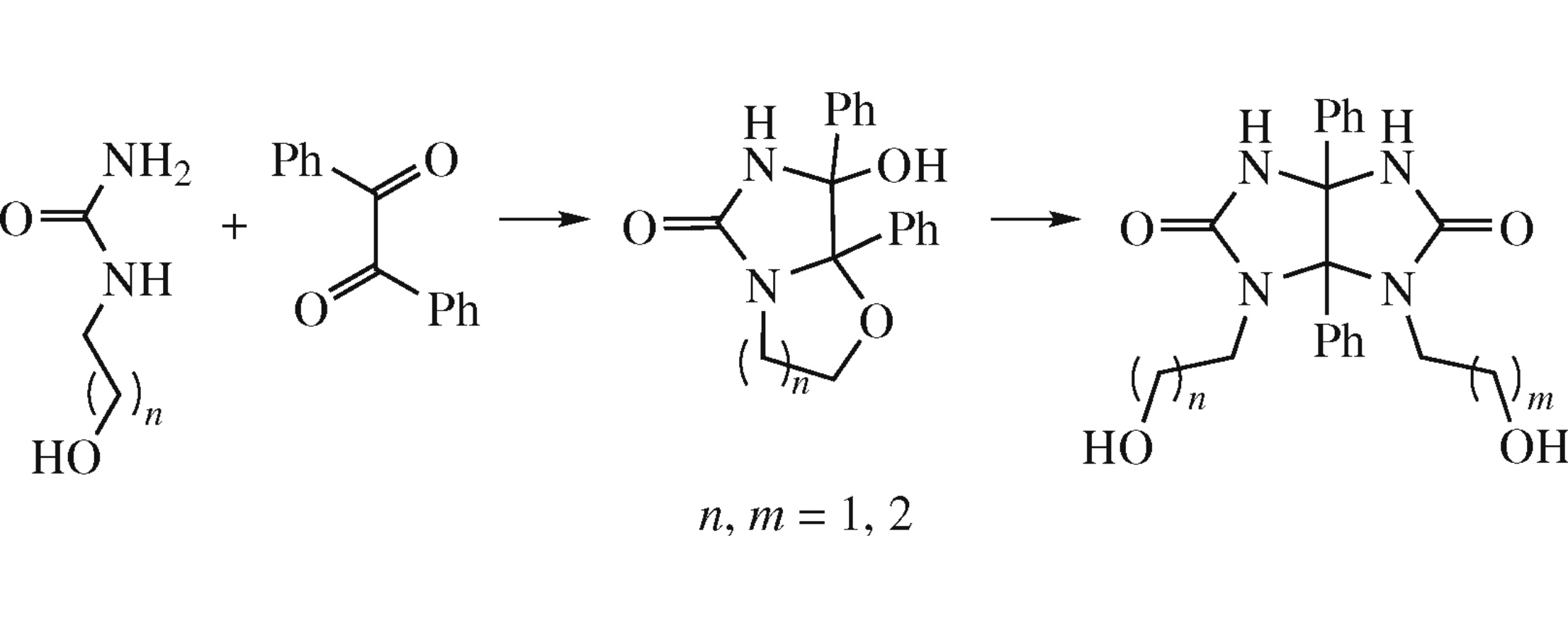
A new synthetic approach to 2,8-disubstituted 1,5-diphenylglycolurils is based on condensation of 1-(hydroxyalkyl)ureas (ureido alcohols) with tetrahydroimidazooxazolone and tetrahydroimidazooxazinone derivatives which were unexpectedly obtained by acid-catalyzed reaction of ureido alcohols with benzil. An X-ray diffraction study of the supramolecular organization of the obtained compounds revealed the chirality of the crystals of achiral glycoluril molecules.
References
1.
10.1016/j.mencom.2014.04.017_bib0005
Mashkovskii
Lekarstvennye sredstva (Drugs), Novaya Volna Moscow,
2013
2.
Ryzhkina I.S., Kiseleva Y.V., Mishina O.A., Timosheva A.P., Sergeeva S.Y., Kravchenko A.N., Konovalov A.I.
Mendeleev Communications,
2013
3.
Kravchenko A.N., Strelenko Y.A.
Mendeleev Communications,
2013
4.

In S., Kang J.
Tetrahedron Letters,
2005
5.

Kang J., In S., Cho S.J.
Supramolecular Chemistry,
2007
6.

Hof F., Craig S.L., Nuckolls C., Rebek Jr. J.
Angewandte Chemie - International Edition,
2002
7.
![Bambus[n]urils: a New Family of Macrocyclic Anion Receptors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Havel V., Svec J., Wimmerova M., Dusek M., Pojarova M., Sindelar V.
Organic Letters,
2011
8.
![Bambus[6]uril as a novel macrocyclic receptor for the cyanide anion](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Toman P., Makrlík E., Vaňura P.
Chemical Physics Letters,
2012
9.

Sun S., Edwards L., Harrison P.
Journal of the Chemical Society Perkin Transactions 1,
1998
10.

Rahimizadeh M., Kam K., Jenkins S.I., McDonald R.S., Harrison P.H.
Canadian Journal of Chemistry,
2002
11.

12.
![Reasons Why Aldehydes Do Not Generally Participate in Cucurbit[n]uril Forming Reactions](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ma D., Gargulakova Z., Zavalij P.Y., Sindelar V., Isaacs L.
Journal of Organic Chemistry,
2010
13.

Stancl M., Gargulakova Z., Sindelar V.
Journal of Organic Chemistry,
2012
14.

Cao L., Meng X., Ding J., Xi H., Wu A.
Journal of Chemical Crystallography,
2011
15.

Wang J., Xiang J., Wu A., Meng X.
CrystEngComm,
2013
16.

Johnson D.W., Palmer L.C., Hof F., Iovine P.M., Rebek J.
Chemical Communications,
2002
17.

Johnson D.W., Hof F., Palmer L.C., Martín T., Obst U., Rebek, Jr. J.
Chemical Communications,
2003
18.
10.1016/j.mencom.2014.04.017_bib0050
Lehn
Supramolecular Chemistry: Concepts and Perspectives,,
1995
19.

Butler A.R., Leitch E.
Journal of the Chemical Society Perkin Transactions 2,
1980
20.
![Glycoluril derivatives form hydrogen bonded tapes rather than cucurbit[n]uril congeners](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Wu A., Fettinger J.C., Isaacs L.
Tetrahedron,
2002
21.

Agami C., Amiot F., Couty F., Dechoux L.
Tetrahedron Letters,
1998
22.

Liao Z.K., Kohn H.
Journal of Organic Chemistry,
1984
23.
![2′,3′,4′,9′-Tetrahydrospiro[cyclohexane-1,1′-(1h)pyrido[3,4-b]indol]-2-ones and their transformations into 2,3,4,4a,5,6,9,14-octahydro-4a-hydroxy-1h,8h-pyrido[3,4-b:2,1-i']diindole-5-carbonitriles and 5-substituted 2,3,4,4a,9,14-hexahydro-4a-hydroxy-1H,8H-indolo[2′,3′:3,4]-pyrido[1,2-c]benzimidazol-6-(5H)ones](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bobowski G.
Journal of Heterocyclic Chemistry,
1981
24.

Metzger C., Kurz J.
European Journal of Inorganic Chemistry,
1975
25.

Kim Y.B., Kim C.S., Lee C.K.
Journal of Heterocyclic Chemistry,
1994
26.

Biltz H.
Berichte der deutschen chemischen Gesellschaft,
1908
27.
![Synthesis of 2-monofunctionalized 2,4,6,8-tetraazabicyclo[3.3.0]octane-3,7- diones](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kravchenko A.N., Maksareva E.Y., Belyakov P.A., Sigachev A.S., Chegaev K.Y., Lyssenko K.A., Lebedev O.V., Makhova N.N.
Russian Chemical Bulletin,
2003
28.
![Synthesis of 2,4-diketo-5-phenyl-.DELTA.5-7oxa-1,3-diazabicyclo[4.4.0]decane and 2,4-diketo-3-phenyl-.DELTA.5-7-oxa-1,5-diazabicyclo[4.4.0]decane](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Smissman E.E., Ayres J.W., Wirth P.J., Abernethy D.R.
Journal of Organic Chemistry,
1972