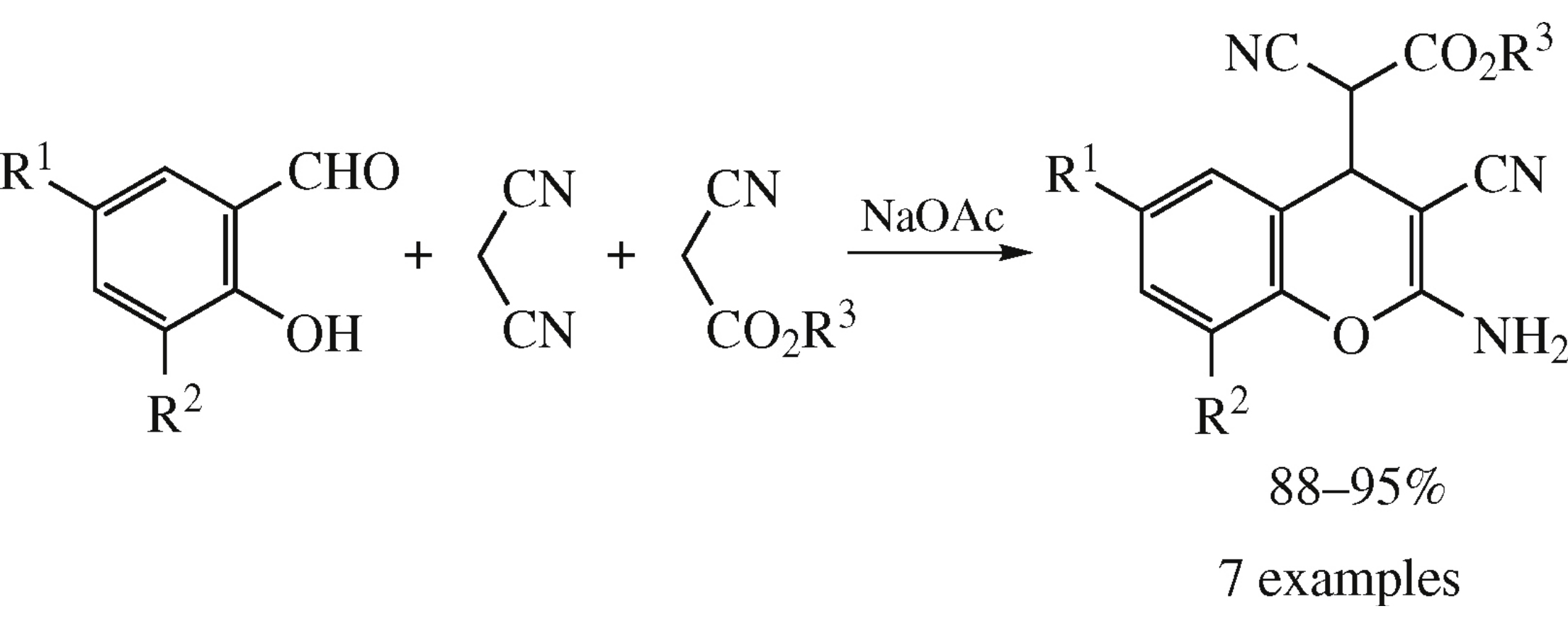
Abstract
Sodium acetate-catalyzed multicomponent assembling of salicylaldehydes, malononitrile and cyanoacetates in water–alcohol mixture (1: 1) at ambient temperature affords alkyl (2-amino-3-cyano-4H-chromen-4-yl)cyanoacetates in 88–95% yields.
References
1.

Orru R.V., de Greef M.
Synthesis,
2003
2.

Weber L.
Drug Discovery Today,
2002
3.
10.1016/j.mencom.2014.04.016_bib0015
Rosen
Pergamon, Oxford,
1991
4.
M. N. Elinson, S .K. Feducovich, T.A. Zaimovskaya, A.N. Vereshchagin and G. I. Nikishin, Russ. Chem. Bull., Int. Ed., 2005, 54, 673.(Izv. Akad. Nauk, Ser. Khim., 2005, 663).
5.

Cleary T., Rawalpally T., Kennedy N., Chavez F.
Tetrahedron Letters,
2010
6.

Elinson M.N., Vereshchagin A.N., Feducovich S.K., Zaimovskaya T.A., Starikova Z.A., Belyakov P.A., Nikishin G.I.
Tetrahedron Letters,
2007
7.

Liu W., Jiang H., Zhu S., Wang W.
Tetrahedron,
2009
8.

2(3H)-Benzoxazolone and Bioisosters as “Privileged Scaffold” in the Design of Pharmacological Probes
Poupaert J., Carato P., Colacino E.
Current Medicinal Chemistry,
2005
9.
![6H-Benzo[c]chromen-6-one derivatives as selective ERβ agonists](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sun W., Cama L.D., Birzin E.T., Warrier S., Locco L., Mosley R., Hammond M.L., Rohrer S.P.
Bioorganic and Medicinal Chemistry Letters,
2006
10.

Stachulski A.V., Berry N.G., Lilian Low A.C., Moores S.L., Row E., Warhurst D.C., Adagu I.S., Rossignol J.
Journal of Medicinal Chemistry,
2006
11.
![New 2-bromomethyl-8-substituted-benzo[c]chromen-6-ones. Synthesis and biological properties](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Garino C., Bihel F., Pietrancosta N., Laras Y., Quéléver G., Woo I., Klein P., Bain J., Boucher J., Kraus J.
Bioorganic and Medicinal Chemistry Letters,
2005
12.

DeSimone R., Currie K., Mitchell S., Darrow J., Pippin D.
Combinatorial Chemistry and High Throughput Screening,
2004
13.

Patchett A.A., Nargund R.P.
Annual Reports in Medicinal Chemistry,
2000
14.

Bonsignore L., Loy G., Secci D., Calignano A.
European Journal of Medicinal Chemistry,
1993
15.

Skommer J., Wlodkowic D., Mättö M., Eray M., Pelkonen J.
Leukemia Research,
2006
16.

Kemnitzer W., Kasibhatla S., Jiang S., Zhang H., Zhao J., Jia S., Xu L., Crogan-Grundy C., Denis R., Barriault N., Vaillancourt L., Charron S., Dodd J., Attardo G., Labrecque D., et. al.
Bioorganic and Medicinal Chemistry Letters,
2005
17.

Kemnitzer W., Drewe J., Jiang S., Zhang H., Wang Y., Zhao J., Jia S., Herich J., Labreque D., Storer R., Meerovitch K., Bouffard D., Rej R., Denis R., Blais C., et. al.
Journal of Medicinal Chemistry,
2004
18.

Gourdeau H., Leblond L., Hamelin B., Desputeau C., Dong K., Kianicka I., Custeau D., Boudreau C., Geerts L., Cai S., Drewe J., Labrecque D., Kasibhatla S., Tseng B.
Molecular Cancer Therapeutics,
2004
19.

Elinson M.N., Dorofeev A.S., Feducovich S.K., Nasybullin R.F., Gorbunov S.V., Nikishin G.I.
Electrochemistry Communications,
2006
20.

Elinson M., Dorofeev A., Miloserdov F., Ilovaisky A., Feducovich S., Belyakov P., Nikishin G.
Advanced Synthesis and Catalysis,
2008
21.
Elinson M.N., Medvedev M.G., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2013
22.
Demchuk D.V., Elinson M.N., Nikishin G.I.
Mendeleev Communications,
2011
23.

Patai S., Israeli Y.
Journal of the Chemical Society (Resumed),
1960
24.
![Synthetic reactions of 2-(2-amino-3-cyano-4H-[1]benzopyran-4-yl)propane-1,3-dinitrile with reactive methylene compounds](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
O'Callaghan C.N., McMurry T.B., O'Brien J.E.
Journal of the Chemical Society Perkin Transactions 1,
1995