Abstract
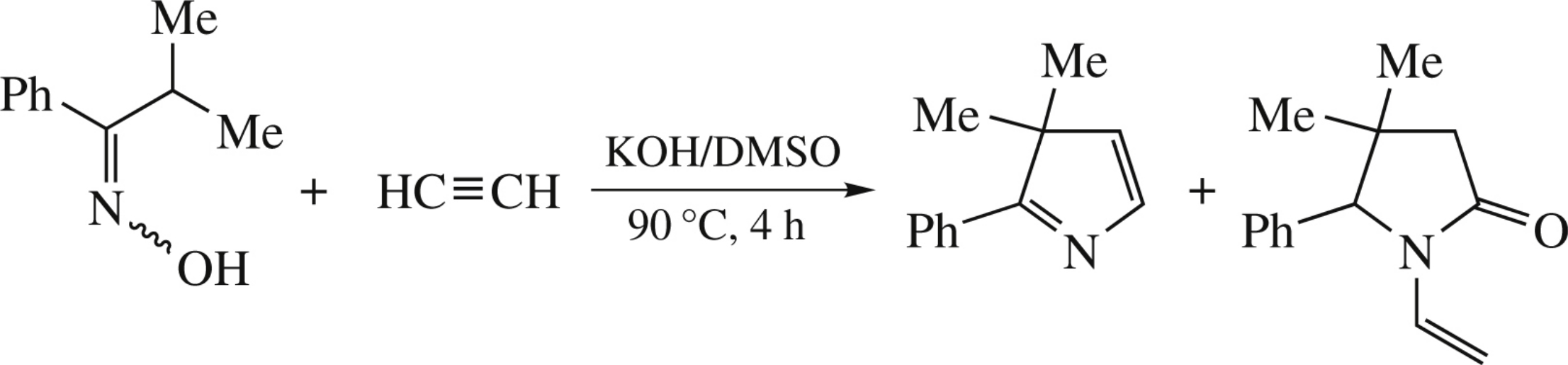
Isopropyl phenyl ketoxime reacts with acetylene in the KOH/DMSO suspension (atmospheric pressure, 90°C, 4h) to afford 3,3-dimethyl-2-phenyl-3H-pyrrole (30%) and 4,4-dimethyl-5-phenyl-1-vinyl-2-pyrrolidinone (5%) that is likely originated from 4,4-dimethyl-5-phenyl-3,4-dihydro-2H-pyrrol-2-ol as the key reaction intermediate.
References
1.
10.1016/j.mencom.2014.03.012_bib0005
Katritzky
New York,
1982
2.
10.1016/j.mencom.2014.03.012_sbref0010a
Sammes
New York,
1990
3.

Trofimov B.A., Shevchenko S.G., Korostova S.E., Mikhaleva A.I., Sigalov M.V., Krivdin L.B.
Chemistry of Heterocyclic Compounds,
1989
4.

Perrin S., Monnier K., Laude B.
Journal of Chemical Research,
1997
5.

Welch K.D., Smith P.L., Keller A.P., Myers W.H., Sabat M., Harman W.D.
Organometallics,
2006
6.
![Isomerization of 3H‐ to 2H‐[1]Benzothieno[3,2‐b]pyrroles and Synthesis of the First Merocyanine Dyes Based on Them](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Shimkin A.A., Shirinian V.Z., Nikalin D.M., Krayushkin M.M., Pivina T.S., Troitsky N.A., Vorontsova L.G., Starikova Z.A.
European Journal of Organic Chemistry,
2006
7.
10.1016/j.mencom.2014.03.012_sbref0015a
Cirrincione
Farmaco,
1992
8.
10.1016/j.mencom.2014.03.012_sbref0015b
Cirrincione
Farmaco,
1996
9.

Padmavathi V., Radha Lakshmi T., Mahesh K., Padmaja A.
Chemical and Pharmaceutical Bulletin,
2009
10.

Merot P., Gadreau C., Foucaud A.
Tetrahedron,
1981
11.

Schmitt G., Nasser B., Dinh An N., Laude B., Roche M.
Canadian Journal of Chemistry,
1990
12.

Depature M., Grimaldi J., Hatem J.
European Journal of Organic Chemistry,
2001
13.
10.1016/j.mencom.2014.03.012_sbref0020d
Xu
Commun.,
2002
14.

Ichikawa J., Sakoda K., Mihara J., Ito N.
Journal of Fluorine Chemistry,
2006
15.
10.1016/j.mencom.2014.03.012_bib0025
Shevchenko
Chem. Heterocycl. Compd,
1985
16.
10.1016/j.mencom.2014.03.012_bib0030
Shevchenko
Chem. Heterocycl. Compd,
1991
17.
10.1016/j.mencom.2014.03.012_bib0035
Shevchenko
Zh. Org. Khim.,
1993
18.
10.1016/j.mencom.2014.03.012_bib0040
Korostova
Chem. Heterocycl. Compd,
1983