Abstract
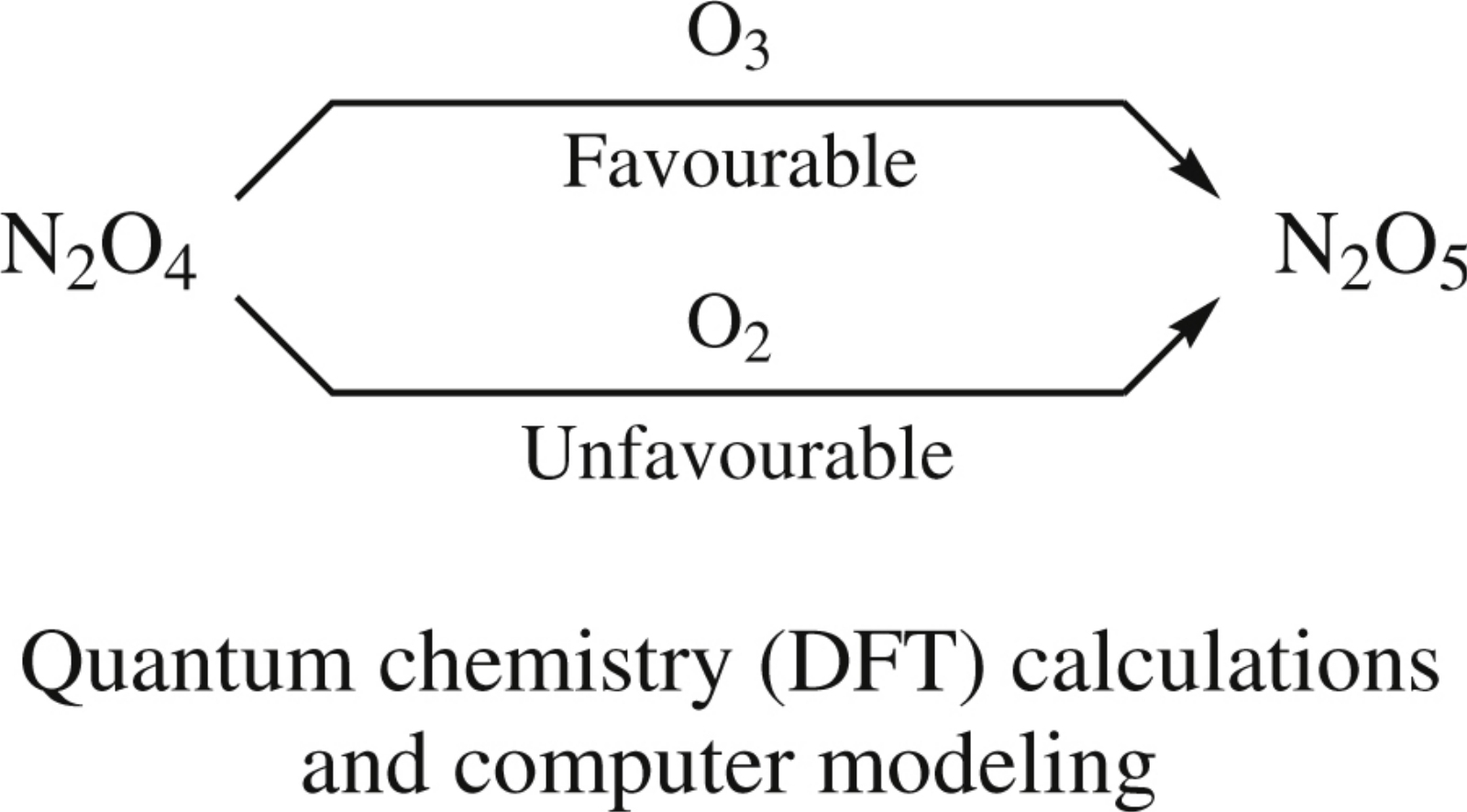
The oxidations of N2O4 to N2O5 with ozone or molecular oxygen were simulated using quantum chemistry (DFT) and computer modeling (Gaussian 98 program package) methods.
References
1.
10.1016/j.mencom.2014.03.009_bib0005
Agrawal
Wiley-VCH,
2011
2.

TALAWAR M., SIVABALAN R., POLKE B., NAIR U., GORE G., ASTHANA S.
Journal of Hazardous Materials,
2005
3.

Philosophical Transactions of the Royal Society of London Series A Physical and Engineering Sciences,
1992
4.

Millar R.W., Philbin S.P.
Tetrahedron,
1997
5.
R. E. Farncomb and G. W. Nauflett, US Patent 6177033, 2001.
6.
10.1016/j.mencom.2014.03.009_sbref0025b
Kuchurov
Russ. Chem. Bull Int,
2010
7.
Synthesis of nitric acid esters from alcohols in a dinitrogen pentoxide/carbon dioxide liquid system
Kuchurov I.V., Fomenkov I.V., Zlotin S.G., Tartakovsky V.A.
Mendeleev Communications,
2012
8.
Kuchurov I.V., Fomenkov I.V., Zlotin S.G., Tartakovsky V.A.
Mendeleev Communications,
2013
9.

Harrar J.E., Pearson R.K.
Journal of the Electrochemical Society,
1983
10.

Logan N.
Pure and Applied Chemistry,
1986
11.
G. Bagg, US Patent 5 181 996, 1990.
12.
G. Bagg, UK Patent 2 229 449, 1991.
13.
G. Bagg, UK Patent 2 245 003, 1992.
14.

Harris A.D., Trebellas J.C., Jonassen H.B., Clemens D.F., Frazier S., Sisler H.H.
Inorganic Syntheses,
1967
15.
10.1016/j.mencom.2014.03.009_sbref0030g
Nitration: Recent Laboratory
ACS, Washington ch. 8,
1996
16.
10.1016/j.mencom.2014.03.009_bib0035
Klapotke
Chemistry of High-Energy Materials,
2012
17.
10.1016/j.mencom.2014.03.009_bib0040
Sarner
New York,
1966
18.
10.1016/j.mencom.2014.03.009_bib0045
Frisch
Gaussian 98, Gaussian, Inc., Pittsburgh, PA,
1998
19.
10.1016/j.mencom.2014.03.009_bib0050
Academic Press, New York,
1964
20.
10.1016/j.mencom.2014.03.009_bib0055
Zakharov
Theor. Exp. Chem., 2007, 43, 66.,
2007
21.

Stirling A., Pápai I., Mink J., Salahub D.R.
Journal of Chemical Physics,
1994
22.

Voegele A.F., Tautermann C.S., Loerting T., Liedl K.R.
Physical Chemistry Chemical Physics,
2002
23.

Zakharov I.I.
Journal of Structural Chemistry,
2011
24.
Yu. A. Ivanov and M. M. Karavaev, RF Patent 2156730, 2000.
25.

Osthoff H.D., Pilling M.J., Ravishankara A.R., Brown S.S.
Physical Chemistry Chemical Physics,
2007
26.

Jitariu L.C., Hirst D.M.
Physical Chemistry Chemical Physics,
2000
27.

Pivina T., Korolev V., Khakimov D., Petukhova T., Ivshin V., Lempert D.
Propellants, Explosives, Pyrotechnics,
2012
28.

Schweitzer C., Schmidt R.
Chemical Reviews,
2003
29.

MULLIKEN R.S.
Nature,
1928
30.
10.1016/j.mencom.2014.03.009_bib0105
Ortenberg
Haifa,
2002
31.

Youmin S., Xiaohua R., Zhaojie C., Guiqin Z.
Journal of Molecular Modeling,
2012
32.

Pakiari A.H., Nazari F.
Journal of Molecular Structure THEOCHEM,
2003
33.
10.1016/j.mencom.2014.03.009_bib0120
Lewis
J. Am. Chem. Soc.,
1916
34.
10.1016/j.mencom.2014.03.009_bib0125
Emanuel
Oxford,
1967