Abstract
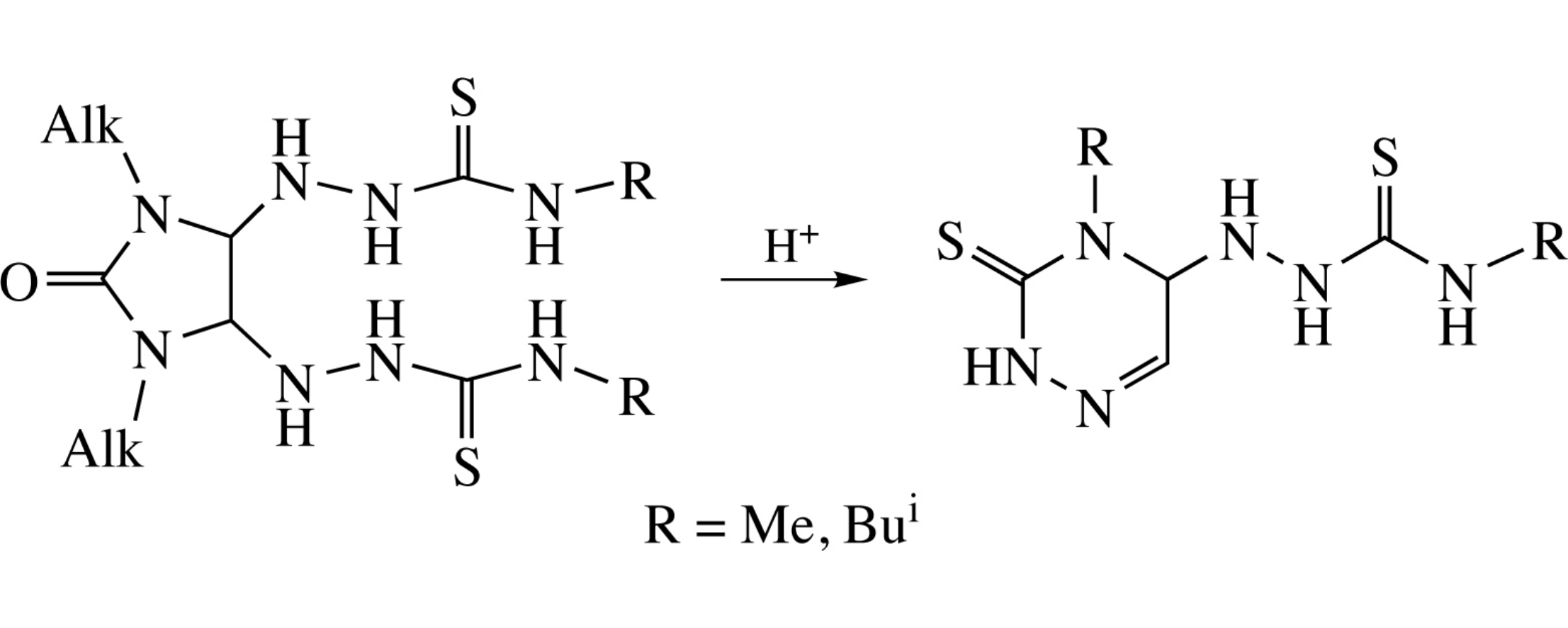
4-Alkyl-5-(4-alkylthiosemicarbazido)-4,5-dihydro-1,2,4-triazine-3(2H)-thiones are obtained either by heating 1,3-dialkyl-4,5-bis-(4-alkylthiosemicarbazido)imidazolidin-2-ones in acidic medium or the reaction of 1,3-dialkyl-4,5-dihydroxyimidazolidin-2-ones with 4-methylthiosemicarbazide.
References
1.

Gazieva G.A., Kravchenko A.N.
Russian Chemical Reviews,
2012
2.

The Wide Pharmacological Versatility of Semicarbazones, Thiosemicarbazones and Their Metal Complexes
Beraldo Heloisa, Gambino Dinorah
Mini-Reviews in Medicinal Chemistry,
2004
3.

Đilović I., Rubčić M., Vrdoljak V., Pavelić S.K., Kralj M., Piantanida I., Cindrić M.
Bioorganic and Medicinal Chemistry,
2008
4.

Huang H., Chen Q., Ku X., Meng L., Lin L., Wang X., Zhu C., Wang Y., Chen Z., Li M., Jiang H., Chen K., Ding J., Liu H.
Journal of Medicinal Chemistry,
2010
5.

Popiołek Ł., Kosikowska U., Dobosz M., Malm A.
Phosphorus, Sulfur and Silicon and the Related Elements,
2012
6.

Nurkenov O.A., Satpaeva Z.B., Kulakov I.V., Akhmetova S.B., Zhaugasheva S.K.
Russian Journal of General Chemistry,
2012
7.

Tenório R.P., Carvalho C.S., Pessanha C.S., de Lima J.G., de Faria A.R., Alves A.J., de Melo E.J., Góes A.J.
Bioorganic and Medicinal Chemistry Letters,
2005
8.

Chipeleme A., Gut J., Rosenthal P.J., Chibale K.
Bioorganic and Medicinal Chemistry,
2007
9.

de Oliveira R.B., de Souza-Fagundes E.M., Soares R.P., Andrade A.A., Krettli A.U., Zani C.L.
European Journal of Medicinal Chemistry,
2008
10.

Rastelli G., Pacchioni S., Sirawaraporn W., Sirawaraporn R., Parenti M.D., Ferrari A.M.
Journal of Medicinal Chemistry,
2003
11.

Sigachev A.S., Kravchenko A.N., Belyakov P.A., Lebedev O.V., Makhova N.N.
Russian Chemical Bulletin,
2006
12.
Gazieva G.A., Vasilevskii S.V., Belyakov P.A., Nelyubina Y.V., Lubuzh E.D., Kravchenko A.N.
Mendeleev Communications,
2010
13.
![A novel synthesis of thioglycolurils by ring contraction of 5,7-dialkyl-3-thioxoperhydroimidazo[4,5- e ]-1,2,4-triazin-6-ones](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Gazieva G., Poluboyarov P., Popov L., Kolotyrkina N., Kravchenko A., Makhova N.
Synthesis,
2012
14.

Gazieva G._., Nelyubina Y.V., Kravchenko A.N., Sigachev A.S., Glukhov I.V., Struchkova M.I., Lyssenko K.A., Makhova N.N.
Russian Chemical Bulletin,
2009
15.

Gazieva G.A., Struchkova M.I., Kolotyrkina N.G.
Chemistry of Heterocyclic Compounds,
2011
16.
Belyakov P.A., Kadentsev V.I., Chizhov A.O., Kolotyrkina N.G., Shashkov A.S., Ananikov V.P.
Mendeleev Communications,
2010