Abstract
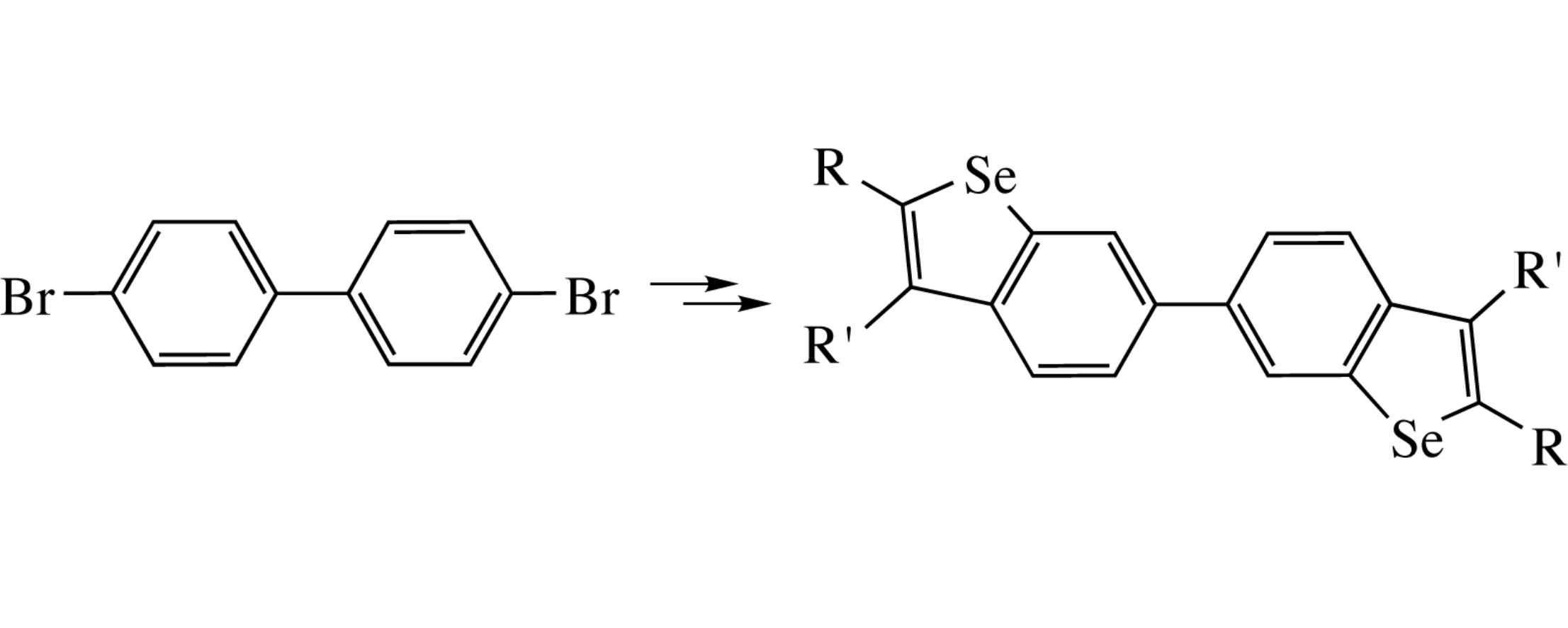
6,6’-Bibenzo[b]selenophenes were prepared from 4,4’-dibromobiphenyl by the Sonogashira coupling with terminal alkynes followed by heterocyclization with SeBr4.
References
1.

Ninomiya M., Garud D.R., Koketsu M.
Coordination Chemistry Reviews,
2011
2.
![Synthesis, structure and cytotoxicity of 3-C, N, S, Se substituted benzo[b]selenophene derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Arsenyan P., Paegle E., Belyakov S., Shestakova I., Jaschenko E., Domracheva I., Popelis J.
European Journal of Medicinal Chemistry,
2011
3.

Shiah H., Lee W., Juang S., Hong P., Lung C., Chang C., Chou K., Chang J.
Biochemical Pharmacology,
2007
4.

Wilhelm E.A., Jesse C.R., Bortolatto C.F., Nogueira C.W., Savegnago L.
Brain Research Bulletin,
2009
5.

Rhoden C.R., Zeni G.
Organic and Biomolecular Chemistry,
2011
6.

7.

Petragnani N., Stefani H.A., Valduga C.J.
Tetrahedron,
2001
8.
T. G. Back, in Organoselenium Chemistry, ed. D. Liotta, John Wiley, New York, 1987, p. 1.
9.

Amosova S.V., Penzik M.V., Albanov A.I., Potapov V.A.
Tetrahedron Letters,
2009
10.

Amosova S.V., Penzik M.V., Albanov A.I., Potapov V.A.
Russian Journal of General Chemistry,
2009
11.

Potapov V.A., Kurkutov E.O., Musalov M.V., Amosova S.V.
Tetrahedron Letters,
2010
12.
10.1016/j.mencom.2013.12.010_sbref0020d
Amosova
Mini-Rev. Org. Chem.,
2010
13.

Braverman S., Pechenick-Azizi T., Gottlieb H., Sprecher M.
Synthesis,
2011
14.

Altomare A., Burla M.C., Camalli M., Cascarano G.L., Giacovazzo C., Guagliardi A., Moliterni A.G., Polidori G., Spagna R.
Journal of Applied Crystallography,
1999
15.
S. Mackay, W. Dong, C. Edwards, A. Henderson, C.J. Gilmore, Stewart, K. Shankland and A. Donald, maXus, Integrated Crystal- lography Software, Bruker-Nonius and University of Glasgow, 2003.
16.
10.1016/j.mencom.2013.12.010_sbref0030c
Sheldrick
2008
17.

Takimiya K., Otsubo T.
Phosphorus, Sulfur and Silicon and the Related Elements,
2005
18.

Pivtsov A.V., Kulik L.V., Makarov A.Y., Blockhuys F.
Physical Chemistry Chemical Physics,
2011
19.

Arsenyan P., Ikaunieks M., Belyakov S.
Tetrahedron Letters,
2007